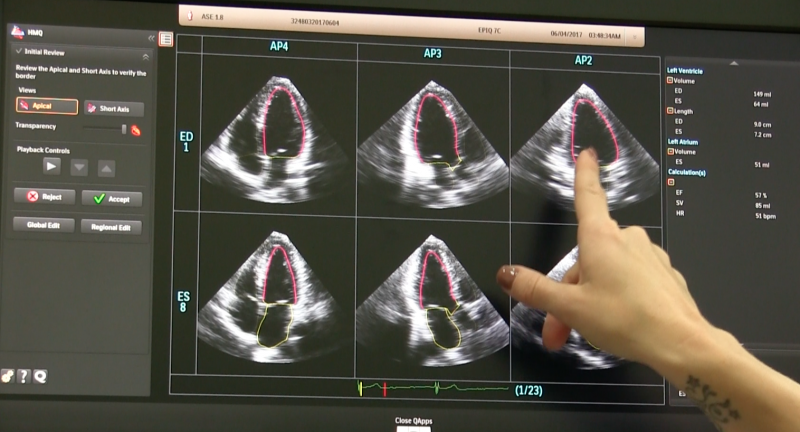
This video educational session, provided in partnership with the American Society of Echocardiography (ASE), is titled ...
Guerbet recently announced that it will phase out sales throughout the world of two products: Hexabrix (meglumine and sodium ioxaglate) and Optimark (gadoversetamide).
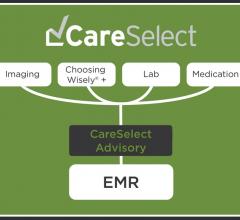
National Decision Support Company announced the full qualification of its CareSelect Platform as a Qualified Clinical Decision Support Mechanism (qCDSM) by the Centers for Medicare and Medicaid Services (CMS). The qualification indicates compliance with the Appropriate Use Criteria (AUC) provisions under the Protecting Access to Medicare Act (PAMA) and the Medicare Access and CHIP Reauthorization Act (MACRA).
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
Leslee Shaw, Ph.D., director of clinical research and professor of medicine at Emory University, Atlanta, and past ...
Matthew Budoff, M.D., FACC, professor of medicine, David Geffen School of Medicine at UCLA, endowed chair of preventive ...
July 18, 2017 — Last-minute defections from two Republican senators early Tuesday ensured the chamber’s version of the ...
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
W. L. Gore & Associates Inc. announced the first patient implant of the Gore TAG Conformable Thoracic Stent Graft with Active Control System after receiving CE Mark last month. The first implant was performed by Prof. Dr. med. Giovanni Torsello and Dr. med. Martin Austermann at St. Franziskus Hospital, Munster, Germany.
Ionizing radiation, such as X-rays, has a harmful effect on the cardiovascular system even at doses equivalent to recurrent computed tomography (CT) imaging, a new study published in the International Journal of Radiation Biology suggests.
July 17, 2017 — The Centers for Medicare and Medicaid Services (CMS) released the 2017 Medicare Trustees Report, the ...
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
The American College of Radiology (ACR) recently revised its authoritative guide for the safe and effective use of contrast media.
LimFlow SA announced enrollment of the first patient in the U.S. feasibility study of the LimFlow Percutaneous Deep Vein Arterialization (pDVA) System. The novel, purely percutaneous device is designed for the treatment of end-stage critical limb ischemia (CLI) when all other revascularization efforts have been exhausted. The first patient was treated on July 5, 2017 at Metro Health - University of Michigan Health by Jihad A. Mustapha, M.D., FACC, FSCAI.
Medical image analysis based on artificial intelligence (AI) employ convolutional neural networks, support vector ...
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
July 17, 2017 — Despite issues with point-of-care INR testing company Alere Inc., Abbott announced this week it is once ...
Intact Vascular Inc. announced the U.S. Food and Drug Administration (FDA) approved an Investigational Device Exemption (IDE) supplemental application to modify the primary endpoint in the Tack Optimized Balloon Angioplasty II Below the Knee (TOBA II BTK) clinical trial from 12 months to 6 months.
A review appearing in the July 18 issue of the Journal of the American College of Cardiology (JACC) discusses current and next-generation implantable hemodynamic monitors. The review particularly examines new approaches focused on the direct measurement of left atrial pressure (LAP), seeking to expand the use of pressure-guided congestive heart failure (CHF) management.

 July 19, 2017
July 19, 2017