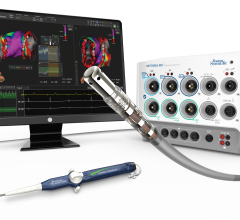
This video, provided by Medtronic, demonstrates the CardioInsight electro-anatomical mapping system. It was cleared by ...
February 1, 2017 — Medtronic received U.S. Food and Drug Administration (FDA) 510(k) clearance for the CardioInsight ...
A new national survey by Orlando Health found that most women are unaware of the age at which heart screenings should begin. The American Heart Association recommends women begin undergoing regular heart screenings at age 20, but the survey found the majority of women, 60 percent, thought screenings didn't need to begin until after age 30, at least a full decade later.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
Heart Test Laboratories Inc. (HTL) has successfully raised $12 million by way of a Common Stock private placement offering. The offering was significantly over-subscribed and provides HTL with a strong financial base entering into 2017. The company expects to commence the international launch of its MyoVista product in the first quarter.
To save the life of an 11-year-old boy, Ann & Robert H. Lurie Children’s Hospital of Chicago has become the first pediatric hospital in the United States to implant the 50cc SynCardia temporary Total Artificial Heart.
Cigna and HeartWell LLP have launched a program to improve quality and cost of care for people diagnosed with chronic coronary artery disease.
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
W. L. Gore & Associates (Gore) announced that the Gore Viabahn VBX Balloon Expandable Endoprosthesis (VBX Stent Graft) has received U.S. Food & Drug Administration (FDA) approval for treatment of de novo or restenotic lesions found in iliac arteries. The approval includes lesions at the aortic bifurcation. This marks the availability of the only balloon expandable stent graft with an indication for the iliac artery.
January 31, 2017 — The American College of Cardiology (ACC) issued a statement against President Donald Trump’s recent ...
January 31, 2017 — Bard Peripheral Vascular Inc. is recalling the Halo One Thin-Walled Guiding Sheath because the sheath ...
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
Annual waste for high-value medical devices continues to grow at a rapid pace. With bundled payments putting increased ...

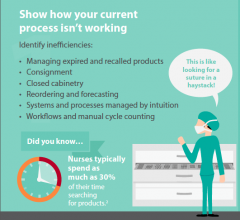
Computerized and semi-automated inventory control system technology offers a more efficient way for cardiovascular departments, especially catheterization and electrophysiology (EP) labs, to track what is on their shelves. These systems can help re-order supplies, track the department’s actual costs per procedure and identify new ways to cut costs.
Intravenous contrast media (typically iohexol or iodixanol) used in computed tomography (CT) does not appear to be associated with chronic kidney disease, dialysis, kidney transplant or acute kidney injury, despite long-held fears to the contrary. The results of the largest controlled study of acute kidney injury following contrast media administration in the emergency department were published online in Annals of Emergency Medicine ("Risk of Acute Kidney Injury Following Intravenous Contrast Media Administration").
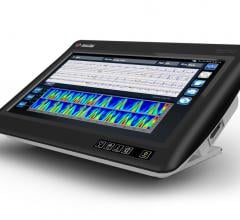
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...

The movement from paper electrocardiogram (ECG) review to electronic ECG management systems in the past decade has ...

Analytics in the past couple years have moved far beyond the simple dashboards used by many cardiology department ...
IBA Molecular has successfully completed its acquisition of Mallinckrodt Nuclear Imaging, announced in August 2016, following the receipt of regulatory approvals.


 February 01, 2017
February 01, 2017