Rivanna Medical announced that it has received U.S. Food and Drug Administration (FDA) 510(k)…
Opsens Inc. announced 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the…
Masimo announced the CE Mark and full market release of MightySat Rx fingertip pulse oximeter…
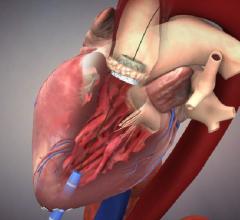
Biosense Webster Inc. announced the launch of the Confidense Module, an innovative technology…
The American College of Cardiology (ACC) Statin Intolerance App is now available to help guide…
Merge Healthcare Inc. announced that it has entered into a strategic partnership with Toshiba …
Summit Imaging announced the release of Summit Imaging Mobile, the mobile version of its…
Cheetah Medical announced that it is now offering the U.S. Food and Drug Administration (FDA)-…
Philips announced the introduction of Lumify, its first app-based ultrasound solution that will…
Medtronic plc announced the U.S. Food and Drug Administration (FDA) approval and U.S. launch of…
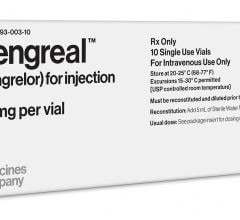
The Medicines Company announced the approval of cangrelor (Kengreal) by the U.S. Food and Drug…
Agfa HealthCare announced that it has received U.S. Food and Drug Administration (FDA) 510(k)…
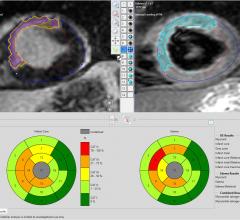
Pie Medical Imaging announced its new release of CAAS MRV, for analysis of the heart to support…
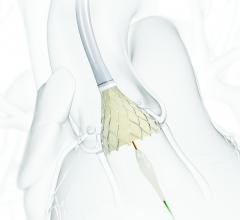
Edwards Lifesciences Corp. announced U.S. Food and Drug Administration (FDA) approval of the…
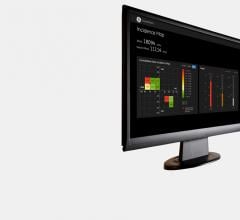
GE Healthcare announced the launch of its new introductory dose management solution, DoseWatch…
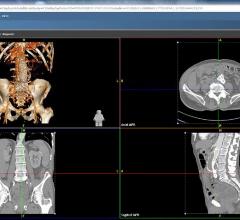
Calgary Scientific Inc. announced the release of ResolutionMD 5.1. The latest software version…
Toshiba America Medical Systems Inc. has received U.S. Food and Drug Administration (FDA)…
In March, Apple announced the launch of ResearchKit, an open source software framework designed…
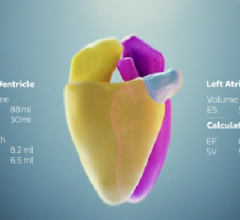
Royal Philips announced the introduction of HeartModelA.I., a new…
June 12, 2015 — Fujifilm Medical Systems U.S.A. Inc. will demonstrate the latest generation of…

 July 14, 2015
July 14, 2015