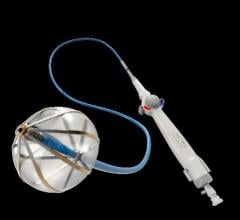
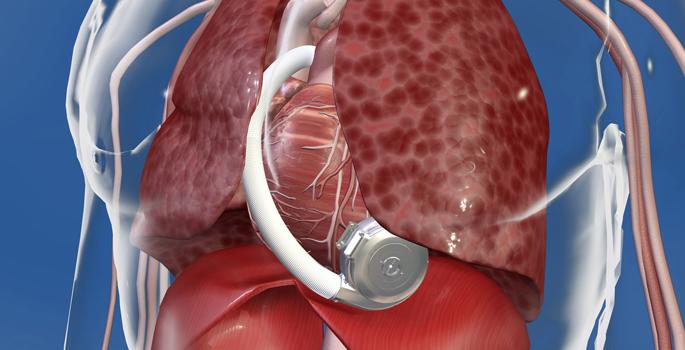
The CardioKinetix Parachute device implant shown deployed in the left ventricle of a heart failure patient. The device helps remodel the ventricle to improve the heart's ability to pump blood more efficiently.
New cardiovascular device therapies for atrial fibrillation (AF) and heart failure (HF) are rapidly evolving with the use of innovative materials and new technologies. While new concepts for cardiac technology are not lacking, successful commercialization is a skill that is also required. Business analysts from the healthcare market research firm Allied Market Research pick out five of the latest innovations in the cardiovascular medical implants segment that should be watched. They feel these technologies have the potential to disrupt the market as new standards for patient care.
HVAD Pump
At present, the HeartWare Ventricular Assist System leads in terms of the implantable ventricular assist devices (VAD) market for patients outside the United States. It received European CE approval from the European Union in 2009, followed by the Australian Therapeutic Goods Administration (TGA) and U.S. Food and Drug Administration (FDA) in 2011 and 2012. The HVAD pump is the benchmark support component of the HeartWare system. To date, more than 11,000 patients with end-stage heart failure have benefitted from the HVAD pump.
As opposed to the competing medical implants available, the smaller pump circuitry of HVAD enables it to be implanted with minimal invasion to provide complete ventricular circulation assistance. The highlight of the design rests within the integrated inflow cannula that eliminates the need for a separate pocket to hold the pump. In addition, once the impeller is powered, there is zero contact between the rotor and the housing. This contactless suspension of impeller reduces hemolysis by providing ample path for circulation.
Read the article HeartWare HVAD System ains FDA Approval for Destination Therapy
HeartMate 3
In line with leading VADs comes the winner of the 2016 Cardiostim-EHRA Europace Innovation Award, the Thoratec HeartMate 3 LVAS. Left ventricular assist device (LVAD) implants across the world have crossed the 50,000 mark. Analysts estimate that about 50,000 to 1 million advanced HF patients in the United States alone are potential candidates for these medical implants.
The HeartMate 3 is capable of pumping up to 10 liters of blood per minute. It was approved with the CE mark in 2015 after a striking 92 percent six-month survival rate during a two-year-long clinical trial. The product faces nonpareil competition from other implants in this segment, being the first commercially approved, fully magnetically levitated LVAD. Attention is now turned to the ongoing MOMENTUM 3 U.S. investigational device exemption (IDE) clinical trial for this device.
St. Jude Medical acquired Thoratec in 2015 to expand its growing heart failure technology portfolio.
Cardialen
The revolutionary Cardialen system aims at restoring the heart’s natural rhythm on AF patients. The small-sized pacemaker implant is delivered through a minimally invasive surgical procedure. Once delivered, it continuously monitors the heartbeat and as soon as AF is detected it emits low-energy electrical pulses that are hardly felt by the patient. There is no requirement for any separate set of delivery devices, and the entire process is performed within a low cost range. The best part — the device can be switched on and off repetitively as per the patient’s convenience, without having to worry about it malfunctioning.
Watchman
Research suggests that AF is the most common form of arrhythmia. At the same time, an estimated 20 percent of strokes occur in patients with AF. In order to reduce the risk of blood clot formation in the left atrial appendage (LAA) of the heart, anticoagulants such as warfarin are widely used. However, the drug itself can cause fatal hemorrhages or increased bleeding due to cuts or ulcers. The Watchman LAA occluder from Boston Scientific gives patients a choice to go off of anticoagulants to a one-time medical device implant. The device can close off the LAA in a transcatheter procedure, and is indicated for patients whose condition is not related to heart valves.
The implant procedure takes about an hour, followed by a 24-hour hospital stay. Within 45 days of the procedure, an individual can stop taking warfarin. The device has had approval for European markets since 2005, and FDA clearance since 2015. The device is approved for commercial use across 70 nations and has been implanted in more than 10,000 patients.
Read the article “Positive Data for Watchman LAA Occluder Real-world Post-FDA Approval Experience.”
Parachute Implant
As the left ventricle remodels due to worsening heart failure, it can become less efficient at pumping, further exacerbating the HF. The transcatheter Parachute structural heart implant from CardioKinetix Inc. helps compensate for the remodeling by separating the portion of the left ventricle that is damaged from the functional part. The subsequent restoration of normal ventricular volume improves the heart’s ability to pump blood more efficiently.
The required size of the implant, which varies by patient, is pre-determined through a computed tomography (CT) scan. The implant procedure takes about an hour and a half. Currently, the device is still under clinical investigation.
While all five of these technologies offer new approaches to AF and HF care, the long-term impact of these technologies will be judged based on clinical outcomes data that is still being collected.
Read the article “Interventional Heart Failure Therapies to Watch.”
Read the article "7 Transformative Medical Technologies to Watch in 2017 and Beyond"
Read the article "Four Paradigm Shift Cardiovascular Technologies to Watch in 2016"
Editor’s note: The author, Abha Mishra, is senior editor at Allied Analytics LLP, part of healthcare market research firm Allied Market Research. She has a large number of publications in the field of biosensors and follows market developments in related market segments. For more information, www.alliedmarketresearch.com

 April 18, 2024
April 18, 2024