September 6, 2023 — The Cardiovascular Research Foundation (CRF) has announced the TCT 2023 late-breaking clinical ...
Balloon Catheter
This channel includes news and new technology innovations for angioplasty balloon catheters (PTA). These are used in arteries with atherosclerotic lesions to compress the plaque expand the artery lumen to reopen occluded or heavily stenosed atherosclerotic lesions. Balloons are often used in combination with a stent to prop the treated vessel segment open. In addition to plain old balloon angioplasty (POBA), this section includes news about drug-coated balloon (DCB), valvuloplasty balloons and specialty cutting balloon.
August 28, 2023 — Prasugrel monotherapy after percutaneous coronary intervention (PCI) with drug-eluting stents is not ...
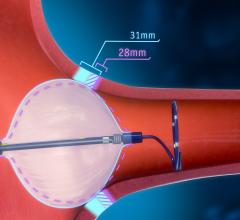
August 9, 2023 — Boston Scientific Corporation announced it has received U.S. Food and Drug Administration (FDA) ...
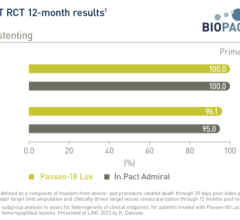
July 14, 2023 — BIOTRONIK announced the one-year subgroup results from the investigator-initiated BIOPACT randomized ...
May 30, 2023 — Twelve-month results from the SELUTION SFA trial have been presented for the first time at the Japan ...
May 30, 2023 — The US FDA, on the 24th of May 2023, granted an Investigational Device Exemption (IDE) approval for Conce ...
May 25, 2023 — Boston Scientific Corporation announced data supporting use of the company's key electrophysiology and ...
May 17, 2023 — Edwards Lifesciences announced that new data from the Benchmark Registry in Europe demonstrated the ...
May 12, 2023 — The US FDA has granted an Investigational Device Exemption (IDE) approval for Concept Medical Inc’s novel ...
May 4, 2023 — The first US patient has been enrolled in the SELUTION4SFA Sirolimus DEB study by Dr. Arthur Lee at the Ca ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
April 28, 2023 — Medtronic today shared additional 36-month data from the IN.PACT AV Access Study, as well as 48-month ...
The European interventional cardiology device market is one of the larger markets in the global space. The market is ...
March 14, 2023 —MedAlliance has announced enrollment of over 1,000 patients in its ground-breaking SELUTION DeNovo coron ...
January 27, 2023 — The first US patient has been enrolled at Medstar Washington Hospital Center in the SELUTION4ISR ...

 September 06, 2023
September 06, 2023