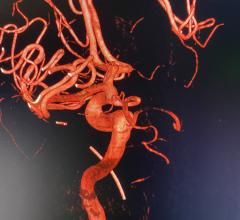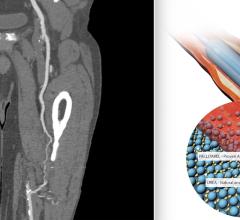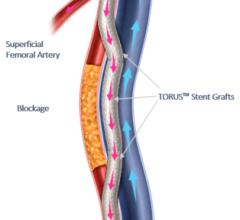
January 17, 2019 — The U.S. Food and Drug Administration (FDA) issued a letter Jan. 17, 2019, to healthcare providers regarding a recent publication that suggests a possible increased risk of death at two years and beyond in patients treated for a type of peripheral artery disease (PAD) with paclitaxel-coated balloons or paclitaxel-eluting stents. The letter was issued in response to a recent publication in the Journal of the American Heart Association1 identifying the risk. It recommends doctors continue to monitor these patients, and discuss the benefits and risks of all available treatment options for patients with PAD.
Paclitaxel-coated balloons and paclitaxel-eluting stents are approved to treat obstructed lesions in arteries of the legs. The authors of the paper analyzed data from previously conducted randomized-controlled trials.
At this time, the FDA believes that the benefits continue to outweigh the risks for approved paclitaxel-coated balloons and paclitaxel-eluting stents when used as indicated. In addition, the agency encourages prompt reporting of adverse events or suspected adverse events experienced with these devices through its MedWatch reporting system.
The FDA is currently evaluating available long-term follow-up data — including from studies that supported approval of the devices and other available data sets — to determine if there are any long-term risks associated with paclitaxel-coated products. The agency is also conducting additional statistical analyses to clarify the presence and magnitude of any long-term risks, and is working directly with manufacturers of these devices to better understand this issue.
The agency said it will communicate publicly as more information becomes available.
For more information: www.fda.gov
Reference
1. Katsanos K., Spiliopoulos S., Kitrou P., et al. Risk of Death Following Application of Paclitaxel‐Coated Balloons and Stents in the Femoropopliteal Artery of the Leg: A Systematic Review and Meta‐Analysis of Randomized Controlled Trials. Journal of the American Heart Association, Dec. 6, 2018. https://doi.org/10.1161/JAHA.118.011245


 March 20, 2024
March 20, 2024