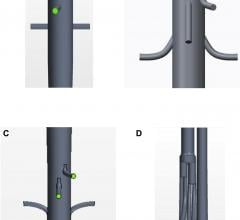
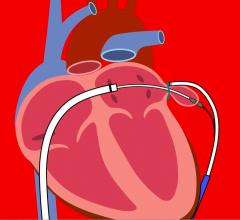
January 6, 2017 — SentreHeart Inc. announced that it has completed the Stage I enrollment milestone in the aMAZE Trial ...
ITN and DAIC Editor Dave Fornell takes a tour of some of the most innovative new technologies being displayed on the ...
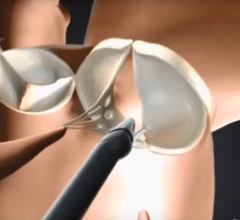
The RECHORD trial allows cardiovascular surgeons to replace damaged string-like tendons, called chordae, through a small incision while the heart is beating. The trial compares outcomes from this minimally invasive procedure to traditional open-heart mitral valve surgery, which requires the chest to be cracked. Specialized imaging is used to place the artificial chordae. PinnacleHealth is one of only 20 sites in the nation and the only hospital in Pennsylvania selected to participate in the trial. Up to 450 patients will be enrolled into the randomized trial.
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
Toshiba Medical announced in November that its Vantage Titan 1.5T/cS Edition magnetic resonance imaging (MRI) system with M-Power V3.6 software received U.S. Food and Drug Administration (FDA) clearance at the 2016 annual meeting of the Radiological Society of North America (RSNA). The new system retains all the patient-friendly features of the Vantage Titan 1.5T MR with added technology to simplify complex cardiac exams.
January 5, 2017 — Whether patients with mechanical heart valves and stents must take blood thinners depends on how ...
Too little sleep takes a toll on your heart, according to a new study presented at the 2016 annual meeting of the Radiological Society of North America (RSNA), Nov. 27-Dec. 1 in Chicago.
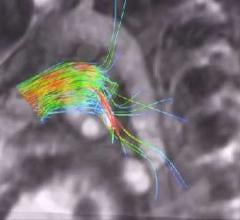
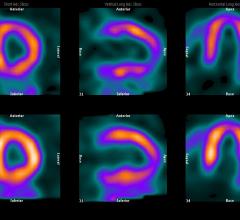
Cardiac positron emission tomography (PET) is growing in popularity among cardiologists because it provides the ability ...
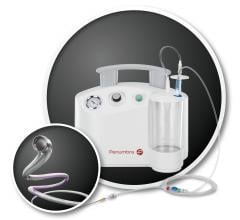
January 5, 2017 — A new report published by Allied Market Research forecasts that the global thrombectomy devices market ...
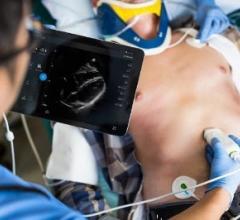
The American Society of Echocardiography (ASE) is in the midst of Echovation Challenge 2017, a competition for its members, and the medical and scientific community at large, to develop innovative solutions in cardiovascular ultrasound technology.
January 5, 2017 – Reacting to a critical shortage of electrophysiology (EP) lab technicians and nurses who treat heart ...
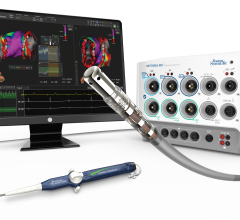
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
At RSNA 2016, Siemens Healthineers unveiled its groundbreaking Compressed Sensing technology, which overcomes a major ...
January 5, 2017 — Working in concert, the American Society of Nuclear Cardiology (ASNC), the Intersocietal Accreditation ...
January 5, 2017 — Abbott announced it completed the acquisition of St. Jude Medical Inc. The transaction provides Abbott ...
Change Healthcare Cardiology Hemodynamics is an integrated hemodynamic monitoring system for monitoring vital signs and ...
In a study published online by JAMA Cardiology, researchers from Stanford University assessed the feasibility of measuring physical activity, fitness and sleep from smartphones. The goal was to gain insight into activity patterns associated with life satisfaction and self-reported disease.
A high-sensitivity blood test could be used to predict which patients are at risk of a heart attack according to new research funded by the British Heart Foundation (BHF).
A new minimally invasive technique for repairing the most common cardiac birth defect in extremely premature newborns can be performed safely with a high success rate in babies as small as 755 grams – about 1.6 pounds – only a few days after birth.

 January 06, 2017
January 06, 2017