The past year saw the release of several new, innovative technologies to improve nuclear imaging in both positron emission tomography (PET) and single-photon emission computed tomography (SPECT).
Since entering the market in 2001, PET/CT has come a long way in combining the benefits of individual PET and CT imaging. New innovations introduced this past year include continuous data acquisition and bed motion, as well as higher image resolutions. The basis of PET/CT is the fusion of anatomical and functional information acquired simultaneously by the hybrid scanner. By capturing anatomy from the CT and combining it with the functional processes observed through PET, physicians have more accurate information with which to diagnose or treat a patient. Furthermore, the resulting image is composed of co-registered scans, meaning they were acquired with the same protocols and with the added benefit of having the patient lying in the same position for both PET and CT modalities. And in a PET/CT, the CT portion provides attenuation correction for the image. H This hybrid modality brings significant advantages to both physicians and patients. Overall scan times are reduced, since the patient is undergoing just one session and not separate PET and CT examinations. It is also convenient for patients who may find going to two separate facilities or a facility twice for two different imaging scans an inconvenience. PET/CT also avoids challenges associated with attempting to fuse a PET image and CT image acquired at different times and possibly at different facilities, improving a physician’s chance of detecting malignant lesions or planning a radiation treatment plan for a patient.
New Systems Debut
When PET/CT emerged a little more than a decade ago, much of its limitations were similar to those faced by individual PET and CT. Today, advances across both modalities are increasing PET/CT capabilities and helping to improve efficiencies for hospitals and institutions that use the combined modality.
Dustin Osborne, Ph.D., an assistant professor in radiology and director of clinical research for the molecular imaging and translational research program at the University of Tennessee School of Medicine, described the state of the PET/CT program during its beginnings in Tennessee. “When I was here 10 years ago as a student, they were averaging about four and a half PET/ CTs a day. And this is a Level-I regional trauma center, so we ought to have been doing more than that,” Osborne said. “We’re actually sitting closer now to about seven PET/CTs per day.”
Osborne uses the latest offering from Siemens Healthcare, the Biograph
mCT Flow PET/CT system first introduced at the 2013 annual meeting of the Society of Nuclear Medicine and Molecular Imaging (SNMMI). Cleared by the U.S. Food and Drug Administration (FDA), it features a new technology, FlowMotion, that moves the patient continuously through the system’s gantry, producing a more robust stream of data that is acquired in a single motion as opposed to previous stop-and-go iterations.
“FlowMotion is simply continuous bed motion,” said Robert Brait, national product manager for PET/CT at Siemens’ Molecular Imaging Division. “Previously you would do a whole-body scan on a stop-and-go scanner, after which you may see that you need to do a high-resolution image of the head and neck or do some type of gating for the chest tube. Now, you can incorporate the high resolution of the head and neck, the gating to correct for motion in the chest and abdomen, and apply some other types of reconstruction. The new technology makes it so the result is a highly customized image, which better illustrates the anatomy and also the pathology occurring in that area.”
Also unveiled in 2013 was the fully digital
Vereos PET/CT system by Philips Healthcare. Released at the 2013 annual meeting of the Radiological Society of North America (RSNA), the Vereos is the first to use digital silicon photomultiplier detectors instead of traditional analog detectors, reportedly doubling the sensitivity gain, volumetric resolution and quantitative accuracy over that of analog systems. The system has received 510(k) clearance from the FDA.
In April, GE Healthcare received FDA clearance and launched its
Q.Clear PET/CT technology, which is designed to provide up to two times improvement in both quantitative accuracy and image quality. Current PET iterative reconstruction, such as time of flight (TOF) and OSEM, forces a compromise between image quality and quantitation. GE said its Q.Clear brings convergence without the need for compromise between quantitation and image quality.
Trends for the Future
The continuous bed motion feature found in Siemens’
FlowMotion technology has received positive feedback thus far from patients and physicians alike, said both Brait and Osborne. In a survey conducted by Osborne and his team at the University of Tennessee, physicians were found to prefer reading studies with data acquired by continuous bed motion versus data collected from step-and-shoot technologies.
“The center part of a PET scanner is the most sensitive and where you get the best resolution,” said Osborne. “With FlowMotion, every part of the patient passes through that central region at some point during the scan, and you get this uniform sensitivity profile across the entire axial extent of the scan. That helps improve some contrast that the physicians see when they’re looking through the axial images.”
The use of this hybrid modality is expected to grow in the coming years, particularly in radiation oncology, which comprises a majority of the volume of PET/CT imaging. “I think that’s an area of PET/CT where we’re going to experience a tremendous amount of growth,” said Osborne.
SPECT Image Quality Improves
The FDA cleared Siemens’
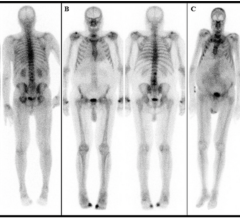
Symbia Intevo SPECT/CT system last September. It is the first system to completely integrate high sensitivity of single-photon emission computed tomography (SPECT) with the high specificity of CT into a single modality, rather than combining as separate overlay images. Siemens is calling the combined modality technology xSPECT.
The system completely integrates data from both modalities, generating high resolution and, for the first time, quantitative images. In traditional SPECT/ CT imaging, the SPECT image has always been reconstructed at a low-resolution matrix — much lower than the CT portion of the exam. As a result, the CT resolution must be downgraded dramatically to the level of SPECT to enable mechanical fusion of the two modalities. xSPECT combines the images into a new, completely integrated single dataset for vastly higher resolution and quantitative images.
New Tracer for Heart Failure
In March 2013, the FDA approved a new indication for GE Healthcare’s
AdreView (iobenguane I-123 injection), the only FDA approved molecular imaging agent to link nerve function in the heart to a patient’s mortality risk. AdreView is approved for the scintigraphic assessment of myocardial sympathetic innervation (cardiac nerve activity) to assist in the evaluation of patients with New York Heart Association (NYHA) Class II or Class III heart failure and left ventricular ejection fraction (LVEF) ? 35 percent.
“Predicting disease progression in heart failure patients can be difficult, and there are currently a limited number of prognostic tools available to help clinicians understand the likelihood for heart failure progression,” said James Arrighi, M.D., associate professor of medicine, Brown University, Providence, R.I. “Now, with AdreView, we have a tool that will provide clinicians with a numeric score to help stratify mortality risk, and may help to promote more informed clinical decision-making.”
Increased myocardial sympathetic activity is a prominent feature of heart failure and is often associated with decline in left ventricular function, worsening heart failure symptoms and sudden cardiac death. This increase leads to a depletion of norepinephrine (NE) storage and uptake. AdreView offers a means to assess neuronal capacity for NE uptake and storage. While current tests look at the effect of the disease on heart muscle and blood flow, AdreView uses the heart to mediastinum ratio to assess the functionality of the sympathetic nerves.
Related Cardiac Nuclear Imaging Content:


 November 12, 2025
November 12, 2025