The key takeaway message from the Society of Cardiovascular Computed Tomography (SCCT) annual meeting in July was that computed tomography (CT) technology is advancing rapidly and there is a strong feeling it will become the pre-eminent cardiac imaging modality.
Experts from top cardiac imaging centers around the country said CT may become a one-stop-shop cardiac imaging test in the near future, eliminating the need for many diagnostic catheterizations, nuclear perfusion scans, stress echo and other tests used to diagnose chest pain or disease progression. They said CT could become the primary gatekeeper to the cath lab for percutaneous coronary interventions (PCI), and help eliminate the high percentage of diagnostic caths performed on patients with normal or non-obstructive disease.
Virtual CT-FFR
A key component of this transformation is a new super-computing technology called CT-fractional flow reserve (FFR), which may offer a noninvasive alternative to catheter-based FFR. Invasive FFR is the current gold standard for determining if a stent is required for a particular lesion. Cardiac CT advocates said this, combined with CT myocardial perfusion imaging, will make CT diagnostics much more accurate. Current cardiac CT relies only on a visual, anatomical assessment of the coronary vessels without any way to qualify the functional impact of atherosclerosis.
“With the ability to now look at functional significance of the lesion, getting away from a purely anatomical based assessment, it is going to be a huge advance,” said Matthew Budoff, M.D., FACC, director, cardiac CT, Harbor UCLA Medical Center, and past president of SCCT.
He explained magnetic resonance imaging (MRI) and nuclear imaging are both the current standards for myocardial perfusion imaging to show if vessel blockages are functionally significant. However, if clinical trials continue to validate CT-FFR and CT perfusion imaging, CT might be able to greatly reduce the need for these additional tests.
There was a large amount of excitement over CT-FFR at SCCT due to the recent release of data from the NXT Trial. This is the third major randomized trial to show significant correlation between CT-FFR and invasive FFR. NXT data and the possible impact CT-FFR may have on the practice of cardiology was discussed in several sessions.
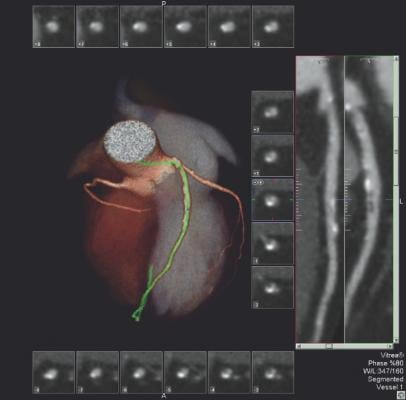
CT-FFR software developed by HeartFlow uses a computational fluid dynamics algorithm to calculate the flow and pressure of blood for the entire coronary vessel tree, said Ricardo Cury, M.D., FSCCT, Baptist Cardiac and Vascular Institute, Miami, Fla. He explained CT-FFR is more sophisticated than invasive FFR, which only provides measurements for a single lesion at a time, in that it provides FFR measurements for all vessel segments at once. The software also takes into account the effect of long diffuse lesions. The system creates a 3-D map of the coronary tree and color-codes segments based on their FFR numbers. The software allows placement of virtual stents and can recalculate the flow virtually to estimate the impact of PCI.
Cury said the DISCOVER-FLOW, DEFACTO and NXT trials tested CT-FFR in more than 600 patients, with excellent correlation with invasive FFR. Data showed marked increases in diagnostic accuracy in CT-FFR vs. CT anatomical imaging alone (81 vs. 53 percent). In comparisons with CT-FFR vs. invasive angiography anatomical assessment, it also was more accurate (86 vs. 71 percent). While the data appears positive, Cury cautioned there still are questions about how calcified lesions or poor CT image quality affect the CT-FFR analysis.
Since CT-FFR uses a global assessment of the coronary tree, some artifacts do not matter because the software looks at flow on either side of these artifacts, said Bjarne Norgaard, M.D., Ph.D., Aarhas University Hospital, Denmark. He began using CT-FFR in April as part of his routine practice for chest pain evaluations and said things like stitching artifacts do not cause a problem. But, heavy calcium bloom or motion blur may cause the software to reject the images for analysis, he said.
This software is in clinical use in Europe, where data from a couple small studies presented at SCCT showed significant cost and time savings over the standard of care.
“I think the data is very convincing,” Norgaard said. “We didn’t really have any other noninvasive imaging modality with this level of accuracy.”
Perfusion Imaging
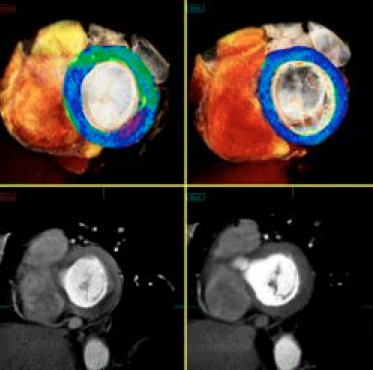
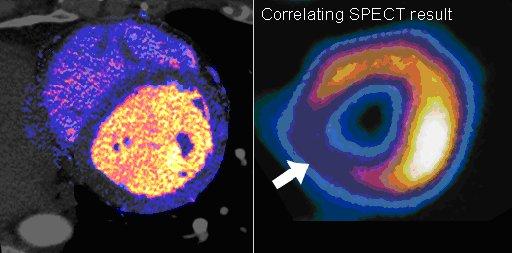
CT perfusion software works by mapping iodine contrast distributions in the myocardium throughout the cardiac cycle. These color-coded maps can be displayed as either bull's eye plots or overlaid on 3-D images of the heart, showing the wash in and wash out of contrast, which is used as a surrogate for blood. Areas of low contrast attenuation correlate with areas of low perfusion due to blocked arteries. Even without attenuation maps, many experienced cardiac CT readers can see shading defects in the myocardium caused by low areas of iodine contrast due to blockages.
While CT perfusion has been available on most CT and advanced visualization vendors’ post-processing software systems for a few years, what had been lacking was clinical data showing how these imaging qualification methods compare to the gold standards of nuclear perfusion imaging and invasive FFR. Budoff said several large, multi-center trials have now been published showing clinical validation. He explained there are specific imaging protocols required for perfusion imaging, making it more complicated than standard CTA, but validation and increasing use has improved many physician’s comfort level with the technology. Many of the patient cases discussed in sessions at SCCT included CT perfusion images and their correlation with invasive angiography.
Spectral Imaging
“I think the next exciting frontier for cardiac CT imaging is spectral CT,” said Guy Weigold, M.D., FSCCT, Medstar Washington Hospital Center, Washington, D.C. “I think we are at the cusp of a whole new area for cardiac imaging.”
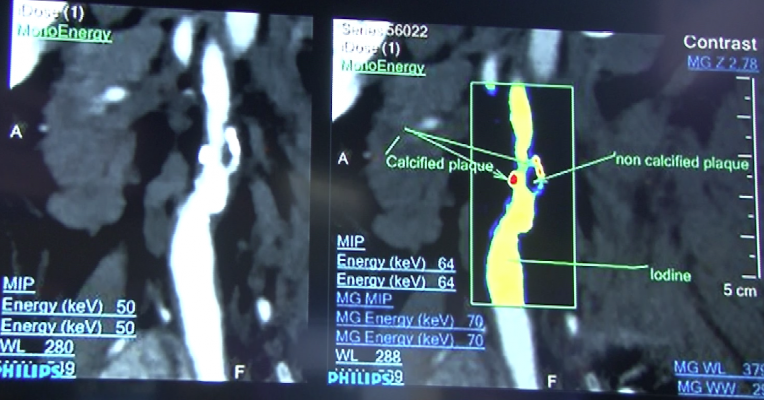
He said spectral CT uses two different kV energies to image the same area of anatomy simultaneously. On a molecular level, some elements have different attenuations at different energies, such as iodine or calcium. This enables the mapping and dialing in or out of these elements from an image to reduce or eliminate calcium blooming and beam hardening artifacts. The technology also allows creation of virtual non-contrast images without the need for a second scan protocol. These methods may help improve image quality and reduce CT radiation dose.
Spectral CT is available on commercially available CT systems. Siemens uses dual X-ray sources and detectors operating at different energies. GE uses a scanner that very quickly alternates kV levels as it scans. Philips developed a unique detector composed of two layers that each detect energies at different kV levels.
Philips Healthcare recently introduced its IQon spectral CT technology. The company hopes to validate it for entering specific atomic numbers off the periodic table of elements to filter images to show concentration of these elements in the body. Weigold said this will allow new ways to characterize tissue or plaque composition based on its chemical makeup.
Spectral imaging also opens the possibility of using an array of new types of contrast agents, including gold, which speakers said has a better absorption rate in tissues than iodine, or compounds that can be used for very targeted biomarkers for specific diseases.
Iodine-based contrast is the current workhorse imaging agent for CT, but it has a short half-life and has been implicated as the culprit in contrast induced nephropathy (CIN). Spectral CT enables the identification of specific element K-edges, which enables imaging of metals using CT. This allows iron and gold to become viable next generation CT contrast agents. Additionally, presenters said metals and other elements can be combined into tailored nanoparticles in sizes between 1-100 microns and in different configurations to target specific receptors.
Plaque Characterization
All advanced visualization and CT manufacturers offer software to characterize plaque composition based on its Hounsfield unit measures. However, the software still requires additional clinical data for validation. Noninvasive CT plaque analysis could have a major impact on evaluating lesions without the need for catheterization, producing data and images similar to the invasive imaging methods of virtual histology intravascular ultrasound (IVUS) or near-infrared spectrography.
“I think we still have some resolution issues, and we need to figure out if studies need to be done at 100 kV or 120 kV and how that impacts the image quality and the measurements,” Budoff said. Additionally, work needs to be done to set a standard for what is a soft, vulnerable, fibrous or calcified plaque at different energies because the numbers can change depending on how the images were acquired. “So, we still need some validation, but things are moving along very rapidly,” he said.
Other issues with CT plaque analysis include the impact of artifacts from calcium blooming and beam hardening artifacts. However, he said IVUS is also very vulnerable to calcium, which prevents imaging of underlying tissue.
Budoff said CT plaque analysis is already starting to take place with pharmaceutical trial evaluations, where it is being used instead of repeated VH-IVUS catheterizations. “I think you will see a very rapid decline in the number of IVUS trials and a very rapid increase in CT-based trials,” he said.
Lowering CT Dose
Budoff said technologies and techniques are available today that allow routine coronary CT at less than 5 mSv. He said using various techniques and newer iterative reconstruction software, his hospital routinely performs cardiac CT at or under 2 mSv using standard 64-slice scanners.
The current standard for most cardiac CT scans is 120 kV, but patient size can greatly vary the amount of kV actually needed to produce diagnostic images, said Brian Ghoshhajra, M.D., director, cardiac imaging, department of radiology, Mass General Hospital (MGH). He said some smaller patients only need 70 or 80 kV. MGH wanted to lower its dose and created new protocols based on body mass index (BMI). He said the basic rule is if a patient’s BMI is below 30, the scanner should be set at 100 kV, and if more than 30, 120 kV.
Ghoshhajra said some BMI can be moved out of the way to enable lower dose scans, as in the case of having a patient move their arms above their head, or moving large breasts out of the imaging field to reduce attenuation issues.
In addition, MGH started recording all its CT doses and publishing the data weekly to show outliers, which he said helped raise dose awareness and helped drive down dose.
Part of the CT dose issue is simply the need to raise awareness about what settings are used on the scanners, how doses at your hospital compares to others, and training techs and setting protocols. An example of how awareness of dose can help lower it was shown in a statewide registry created by Gilbert Raff, M.D., director of advanced cardiovascular imaging, Beaumont Health Systems, Royal Oak, Mich. He helped create the Advanced Cardiovascular Imaging Consortium (ACIC), a statewide quality improvement registry funded by Blue Cross Blue Shield of Michigan. During the first month, the median dose was 25 mSv, but declined over a nine month period by 48 percent to an average dose of 13.1 mSv.1
Video Coverage From SCCT
Reference:
1. ?Gilbert Raff; Kavitha Chinnaiyan; Aiden Abidov, et al. “Marked Radiation Dose Reduction in a Statewide Coronary CT Quality Improvement Registry.” Circulation. 2008;118:S_936.


 February 02, 2026
February 02, 2026