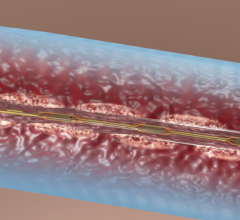
The Bayer Healthcare AngioJet system uses jets on the side of the catheter to help break up thrombus and infuse thrombolytic agents.
Catheter-based thrombectomy systems are increasingly being used to remove blood clots from vessels inside the body, often as an adjunct therapy to systemic pharmaceutical thrombolytics. These systems are used in veins, as in the case of deep vein thrombosis (DVT), and in arteries, in the case of pulmonary embolism (PE), ST-elevated myocardial infarction (STEMI) and ischemic stroke. The systems can also quickly clear dialysis arteriovenous grafts, which are prone to thrombus formation.
Invasive thrombectomy is an interventional area many cath labs are expanding into to boost patient volumes. Traditionally DVT and other non-coronary clot-based conditions were treated with drugs (lytics) to break down the clot over hours or days. However, many centers are now looking at more aggressive and targeted treatment of clots using catheter-based therapies.
There are three types of catheter thrombectomy devices on the market — aspiration catheters to vacuum the clot out of the vessel, lytic deliver catheters to infuse the clot with targeted thromobytic drug treatment, and mechanical thrombectomy systems that break up the clot. Systems on the market may combine one or more of these attributes in their systems. There are pros and cons to each of these approaches.
Aspiration Catheters
• Advantages: Offers a simple, low-cost option for removing small, fresh, soft thrombus from coronary arteries. Current STEMI guidelines recommend use of these devices. These catheters are capable of rapid, single-session flow restoration in the setting of small thrombus.
• Disadvantages: Larger and more organized thrombus can overwhelm and clog the small aperture of a manual aspiration catheter. Recent studies using optical coherence tomography (OCT) imaging have shown that thrombectomy may leave some thrombus behind, which may negatively impact stent placement and clinical outcomes. Manual aspiration is more likely than mechanical methods to leave residual thrombus. Additionally, recent studies have shown larger profile aspiration catheters have difficulty crossing lesions.
Lytic Delivery Catheters
• Advantages: Catheter directed thrombolysis (CDT) is the localized delivery of lytic via catheter to dissolve thrombus to restore vascular flow. Lytic therapy offers improved outcomes versus standard anticoagulation therapy.
• Disadvantages: Lytic alone is often not fast enough to resolve a critical coronary blockage as found in STEMI or in restoring flow in peripheral vasculature. In peripheral cases, a lytic-only treatment may require extended stays in the ICU and frequent angiographic revisualizations to check on the progress. For large thrombus burden, there may be a significant systemic dose of lytic before the blockage is fully resolved. Systemic and higher doses of lytic delivery also increase the risk of bleeding.
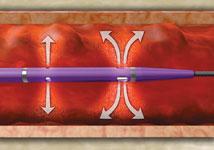
Mechanical Thrombectomy
• Advantages: Using mechanical thrombectomy to break up and eliminate the venous thrombus early offers benefits in maintaining venous valve function, reducing the risk in developing deep vein thrombosis (DVT) and ultimately post-thrombotic syndrome (PTS). It also removes a higher percentage of clot than manual methods and can restore flow in significantly less time than lytic alone. Mechanical thrombectomy is capable of single-session flow restoration. In the setting of large thrombus in STEMI, mechanical thrombectomy has shown to reduce thrombus burden and improve reperfusion and clinical outcomes as compared to standard percutaneous coronary intervention (PCI) and manual aspiration.
• Disadvantages: Mechanical thrombectomy requires more endovascular skill and technique. In some patients, in specific anatomy, it may induce temporary bradycardia and long runtimes in flowing fields. Without proper patient hydration, mechanical thrombectomy may create temporary hematuria/hemoglobinuria.
Pharmacomechanical Thrombectomy Systems
Utilizing mechanical thrombectomy in conjunction with the ability to infuse medication through the catheter directly into the thrombus site can often reduce the duration and dose of medication. This often provides resolution of the blockage while reducing the overall systemic dosage associated with a CDT approach and the time patients may need to be on lytic therapy. The disadvantages are the same as mechanical thrombectomy systems.
Industry Changes
Maquet acquired Atrium Medical in 2012, adding its Clearway infusion catheter to its product line. Maquet will also benefit from Atrium’s continuing development of other minimally invasive thrombectomy devices. The INFUSE-MI trial examined the use of the Clearway device to reduce infarct size in myocardial infarction patients by targeted delivery of lytic therapy to the culprit coronary artery segment.
The Atrium ClearWay RX catheter was used in the INFUSE-AMI trial, which was published in March 2012. The study found STEMI patients who had the antiplatelet drug abciximab delivered directly to the culprit lesion via the catheter had better outcomes. The same study also found clot aspiration did not significantly reduce the damage to the heart muscle.[1]
New Opportunities to Expand Cath Lab Volume
The Vascular Disease Foundation (VDF) said DVT occurs when a blood clot forms in the deep veins, usually of the pelvis or leg. It can be fatal if a clot breaks free from the leg veins and travels through the heart and lodges in the lungs, creating a pulmonary embolism (PE). PE causes between 100,000 and 180,000 deaths per year in the United States, according to the VDF. DVT can also cause permanent damage to the veins, called post-thrombotic syndrome (PTS) or chronic venous insufficiency. PTS symptoms include long-term leg pain, swelling and difficulty walking, and can lead to changes in skin color and open leg ulcers, significantly impacting patients’ quality of life.
With declining cardiac patient volume, many cath labs are looking at other types of procedures to boost the caseload, and venous interventions are becoming more popular. This trend was very noticeable at the annual Transcatheter Cardiovascular Therapeutics (TCT) meeting in October 2012, where there was a definite uptick in sessions and vendors on the show floor addressing venous interventions.
The National Institutes of Health is sponsoring the ATTRACT study, a $10 million, multicenter randomized trial that is being performed in 50 hospitals nationwide. The study is expected to be a landmark trial examining the impact of intervention therapies compared to anticoagulant therapy alone in treating DVT to prevent PTS and improve long-term quality of life. The study is still recruiting patients. The interventional arm includes intra-thrombus delivery of recombinant tissue plasminogen activator (rt-PA) into the DVT over a period of up to 24 hours. Three methods of rt-PA delivery will be used, including the Trellis-8 Peripheral Infusion System, the AngioJet Rheolytic Thrombectomy System and catheter-directed rt-PA infusion via a multi-sidehole infusion catheter.
Comparison Chart
This story was an introduction for a comparison chart of clot busting technologies that ran in the Jan/Feb 2013 issue of DAIC. To access this chart, click on the "Comparison Charts" tab at the top of this webpage. Participants included:
• Argon Medical (Rex Medical) — www.argonmedical.com
• Bayer — interventional.bayer.com
• Control Medical — AspirationMedical.com
• Ekos Corp. — www.ekoscorp.com
• Medronic Inc. — www.medtronic.com
• Vascular Solutions — www.vasc.com
Companies that declined to participate include Covidien, Edwards Lifesciences, Maquet and Spectrinetics.
Reference:
1. Gregg W. Stone, Akiko Maehara, Bernhard Witzenbichler, et al. “INFUSE-AMI trial: A 2x2 Factorial, Multicenter, Prospective, Randomized Evaluation of Intracoronary Abciximab and Aspiration Thrombectomy in Patients Undergoing Primary PCI for Anterior STEMI.” JAMA. Published online March 25, 2012.


 November 21, 2022
November 21, 2022