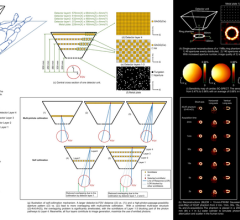
Lantheus is developing flurpiridaz (an 18-F labeled PET agent) to help detect smaller occlusions. It has a half-life of two hours and is expected to undergo final FDA review in 2014.
There are currently three major trends in cardiac nuclear perfusion imaging technology: advances in detector hardware, improved software and new radiotracers. These advances have enhanced the diagnostic performance of both single photon emission computed tomography (SPECT) and positron emission tomography (PET) perfusion imaging. To hear the latest on these trends, Diagnostic and Interventional Cardiology (DAIC) spoke with Marcelo Di Carli, M.D., FACC, FAHA, co-director, cardiovascular imaging; section chief, nuclear medicine, Brigham and Women’s Hospital and associate professor of radiology, Harvard Medical School. He also serves as a member on the Society of Nuclear Medicine (SNM) Cardiovascular Council Board of Directors.
New SPECT Technology
“One of the major trends we have seen over the past three years or so is the development of new dedicated cameras for cardiac-specific features,” Di Carli said.
He said these features include higher sensitivity with the use of new types of detectors, smaller field-of-view, reduced dose and better designs for increased patient comfort. In addition, new software has greatly improved the counting and accuracy in SPECT perfusion imaging.
Among the new cameras are the D-SPECT from Spectrum Dynamics and GE Healthcare’s Alcyone Technology family of SPECT detectors. Both use a new cadmium zinc telluride (CZT) detector, which is more sensitive than previous types of gamma cameras. They directly convert photons into digital signals, eliminating the need for crystals and photomultiplier tubes. Di Carli said this has greatly reduced the size of these systems and the footprint.
The improved image quality provided by CZT prompted GE Healthcare in November 2010 to acquire OMS, a subsidiary of Orbotech, which manufactures GE’s CZT detectors. These detectors are used on both the Discovery NM 530c and Discovery NM/CT 570c.
“These solutions allow you to deal with more noise associated with lower-dose scans and to improve spatial resolution.” Di Carli said. “It is possible to reduce scan times and patient dose.”
These detectors, combined with new software, have greatly improved photon counting statistics, which Di Carli said allows radiologists and cardiologists to adjust the tracer dose and the duration of scans. This has contributed to reduced patient radioactive tracer dose and cut exam times. The increased sensitivity of the GE and Spectrum Dynamics scanners allows perfusion exams to be completed in two to six minutes, which Di Carli said is a significant improvement over current technologies requiring 12 to 20 minutes. SPECT patient radiation dosage averages 11 to 12 mSv, but the more sensitive detectors and improved software have reduced this to 5 mSv. “We are talking about a reduction of more than 50 percent,” Di Carli said.
Another camera advancement includes Digirad’s Cardius X-ACT camera. It was among the first SPECT systems that combined solid-state detectors, a rapid imaging detector geometry, a low-dose volume computed tomography (CT) attenuation correction approach and reconstruction software combined into an upright imaging unit.
Di Carli said Siemens’ IQ-SPECT system also offers advances, with a new collimator design and improved reconstruction techniques to allow greater detector sensitivity and improved spatial resolution.
The better collimation and the smaller field-of-view in newer cameras specifically designed for cardiac imaging also reduce tracer signal from surrounding, non-cardiac tissue, which previously obscured the view of the heart, Di Carli added.
Software Quantification Advances in PET
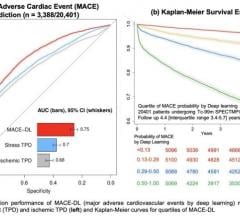
Di Carli said advances in PET post-processing software are helping quantify the flow of blood in cardiac tissue to estimate coronary flow reserve. He explained the software has become accurate enough to show triple-vessel disease involvement and better stratify the risk in intermediate-risk patients.
“In the past, quantifying blood flow was really just used as a research tool,” Di Carli said.
A Major Blow to Cardiac PET
The primary tracer used for U.S. PET cardiac perfusion exams is Bracco’s CardioGen-82 generator, which was pulled off the market in July. The U.S. Food and Drug Administration (FDA) and Bracco pulled the tracer after two patients who had cardiac PET exams set off U.S. border crossing radiation detectors due to strontium breakthrough from use of the agent.
“(CardioGen-82) represents 95 percent of all PET cardiac exams in the United States,” Di Carli said. “It’s a huge setback, because PET has demonstrated a clear increase in accuracy over SPECT, so it is really a blow to patients.”
Di Carli said the only other option for PET cardiac perfusion imaging is N-13 ammonia, which requires an on-site cyclotron to produce. However, a new cardiac PET agent is in development and has the added benefit of a much longer half-life.
Advances in Radiotracers
While PET’s image quality is better than SPECT, a major issue has been the extremely short half-life of its radiotracers (about 75 seconds). However, this critical time element may be offset with a new agent being developed by Lantheus. The company recently entered Phase III development of flurpiridaz (formerly known as BMS747158). This fluorine-18 (18-F) labeled agent binds to the mitochondrial complex one inhibitor. Di Carli said it allows tracking of myocardial blood flow in a linear fashion, which can help detect smaller occlusions. In addition, since the agent is 18-F based, it has a half-life of two hours. He said the product is expected to undergo final FDA review in 2014.
“It will be a huge improvement for cardiac PET,” Di Carli said.
GE Healthcare is developing a new SPECT agent, iodine 123 (I-123) metiodobenzylguanidine (MIBG), for imaging the sympathetic nervous system of the heart. The sympathetic nervous system is responsible for regulating myocardial blood flow, heart rate and the heart’s contraction performance. In patients with coronary artery disease, heart failure or arrhythmias, sympathetic innervation is altered and measurements of receptor density in the myocardium can be used to make a diagnosis and determine prognosis. I-123 MIBG is an analog of norepinephrine, which acts as a false neurotransmitter stored in the nerve terminal and can help measure receptor density. Di Carli said the agent will help identify patients with malignant ventricular arrhythmias and allow risk assessment of patients at high risk for cardiac mortality.
The MIBG tracer is under final review by the FDA and Di Carli said it could receive market clearance before the end of 2011. GE already sells the tracer under the trade name AdreView for the detection of primary or metastatic pheochromocytoma or neuroblastoma cancer tumors.
Reimbursement Issues
Six years ago there was a big trend in nuclear imaging toward adopting smaller physician office-based SPECT imaging systems. However, that trend was blunted by changes in reimbursements, primarily as a way to stop a large increase in practice self-referrals. Over the past 15 years, Di Carli said there was an 80 percent expansion in outpatient SPECT imaging, but much of that imaging is reverting back to hospitals due to reimbursement.
“Outpatient practices have been hit hard by the reimbursement issues and many practices have sold imaging back to hospitals and now only interpret the images,” Di Carli said.
With the Centers for Medicare and Medicaid (CMS) interested in reducing healthcare costs and wanting to bundle more payment, Di Carli said it is unlikely this trend will reverse. He said hospitals would likely remain the chief nuclear imaging providers in the foreseeable future.

 June 05, 2023
June 05, 2023