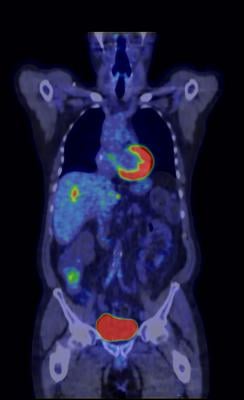
April 8, 2014 — GE Healthcare received U.S. Food and Drug Administration (FDA) 510(k) clearance of its Q.Clear technology, which is designed to provide up to two times improvement in both quantitative accuracy and image quality in PET/CT imaging.
Over the last decade, positron emission tomography (PET) image reconstruction technology has been designed to provide better image quality, reduced acquisition time and lower injected dose. Current PET iterative reconstruction technologies, such as time of flight (TOF) and OSEM, force a compromise between image quality and quantitation. GE Healthcare said its Q.Clear technology brings convergence to PET imaging without the need for compromise between quantitation and image quality.
"Q.Clear is a major step forward because it can give us a consistent and reliable measurement when determining whether the current course of a patient's cancer treatment is effective," said Dr. Gustav von Schulthess, nuclear medicine chair at University Hospital Zurich. "It will give the oncologist more confidence because if a change of therapy is needed, you want accurate information early on to best adjust treatment for the patient."
Clinicians not only want the ability to detect smaller lesions, but the ability to determine earlier whether the patient is responding to current treatment. Q.Clear aims to help in both areas by providing better quantitation accuracy for truly personalized medicine. Coupled with Q.Suite, which introduced the importance of eliminating variability such as respiratory motion, Q.Clear can enable clinicians to assess treatment response accurately.
"We know that approximately 70 percent of cancer patients don't always respond to their initial course of treatment," said Steve Gray, president and CEO of GE Healthcare MICT. "If we can give clinicians an accurate, reliable, and faster tool to confirm that a change in treatment is needed, the patient will benefit greatly. For example, PET/computed tomography (CT) can help clinicians determine whether chemotherapy is working in fewer cycles, saving patients unnecessary procedures. Q.Clear gives clinicians the ability to help make that determination."
GE said the system may not be available for sale in all regions. For countries that require CE marking, Q.Clear is not yet CE marked.
For more information: www3.gehealthcare.com

 November 17, 2025
November 17, 2025 





![Phase III clinical trial of [18F]flurpiridaz PET diagnostic radiopharmaceutical meets co-primary endpoints for detecting Coronary Artery Disease (CAD)](/sites/default/files/styles/content_feed_medium/public/Screen%20Shot%202022-09-13%20at%203.30.13%20PM.png?itok=2w6OoNd6)
