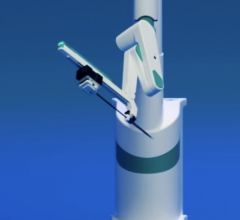
A rendering of the CorPath Remote Control Catheterization system
Like medieval knights in battle, stent-wielding cath-lab doctors arm themselves with heavy and cumbersome lead garments to avoid repeat exposure to radiation when performing traditional angiographic procedures.
“It’s a double-edged sword,” explained Rafael Beyar, M.D., director of Invasive Cardiology at Rambam Medical Center in Haifa, Israel. “The heavier these aprons are, the more protective they are. Some of my friends wear two of them because of fear of exposure.”
But instead of increased cancer risk, these doctors end up with back pain and disc problems.
“Some reports estimate that 80 percent of cardiologists have back problems, and many of them are forced to quit clinical work at the age of 50 or 55 because of spine disease,” he said. “It’s sometimes even bigger than the cancer problem.”
Like many others in his line of work, William T. Katsiyiannis, M.D., is familiar with being weighed-down on the job.
“Long-term orthopedic consequences of standing in lead aprons have resulted in the premature ending of many careers,” said the electrophysiologist with Minneapolis Heart Institute at Abbott Northwestern Hospital. “These procedures require long periods of standing while wearing lead aprons and also result in high fluoroscopy times.”
But it appears that with a little help from robotics, the days of the “doctor in shining armor” are becoming history.
Blazing its Own CorPath
Dr. Beyar is cofounder of Corindus Corp., one of a few companies that are developing ways to conduct these procedures remotely. Corindus’ CorPath is a system that allows physicians to advance and torque coronary guide wires and stent systems in a patient’s artery while eliminating the need for protective gear.
The company completed its animal and first-in-man (FIM) trials approximately one year ago and is now working with the FDA to finalize its Investigational Device Exemption (IDE) to start human clinical trials in the U.S. The CORRECT, or Coronary Remote Control Catheterization Trial, will be carried out at five sites within the Boston-Washington D.C. corridor and will be lead by Ron Waksman, M.D., of Washington Heart Center in Washington, D.C.
Tal Wenderow, Corindus’ CEO, says CorPath will use the Stereotaxis Magnetic Navigation System as the predicated device and anticipates FDA clearance in late 2007. He emphasizes that it may be the only commercially available remote control system with an indication for percutaneous coronary intervention (PCI).
“One of CorPath’s distinct features is its compatibility with conventional 0.014-inch angioplasty instruments,” Wenderow said.
The system is comprised of a bedside unit and a physician workstation. The bedside unit is an electromechanical device located at the edge of the operating table and houses guide wire and stent system manipulators. It is positioned near the patient at the femoral access site.
“Patients are positioned on the table in the traditional way,” Dr. Beyar said. “They are awake and everything is done the same way as it would be with catheterization.”
The bedside unit is designed to mechanically maneuver guide wires and stent systems with various motion mode options. Devices can be maneuvered rotationally, axially or simultaneously, and in precise controllable increments. The motion mode is specified by the physician at the physician workstation.
“The operation is done with a joystick by the physician, away from the source of X-ray radiation,” Dr. Beyar said. “It can be two meters away behind a shield, in another room, in another town or in another continent.”
Lighting up a Stenosed Bifurcation
The Stereotaxis Magnetic Navigation System has been used by Dr. Katsiyiannis since the spring of 2005. He says learning to tell a computer how to move a catheter is “different,” but extremely useful once learned.
“Having performed thousands of procedures by hand, learning a totally new method of navigation can at first slow things down,” he said. “However, one can learn fairly quickly.”
The system is designed to enable physicians to complete complex interventional procedures by using computer-controlled, externally applied magnetic fields that govern the motion of the working tip of the catheter or guide wire.
In fact, the system’s endoluminal view was recently used in successfully navigating a triple magnet-tipped .014 RF guide wire through a stenosed bifurcation — the narrowing of a main vessel at an angled branch — with minimal contrast use. The results were presented at TCT 2006.
Dr. Katsiyiannis said the system’s procedural benefits include increased precision of catheter movements, improved stability of the catheter in difficult locations and computer memory of difficult anatomic locations.
“Additionally, the Stereotaxis catheter is very soft, making the perforation risk much lower,” he added. “This allows for better ability to navigate in complex cardiac chambers.”
Patient feedback has been “universally positive,” he said. “Plus, a technology that allows one to sit down, without wearing lead, and to avoid practically all radiation exposure is invaluable.”



 January 27, 2026
January 27, 2026