Use of
therapeutic hypothermia is used to prevent neurological damage in patients who suffer sudden cardiac arrest (SCA), and there are many advocates suggesting a similar approach can help improve outcomes in ST-elevated myocardial infarction (STEMI) patients.
Clinical data shows lowering a patient’s body temperature from the normal range of 37-38 C to a mild hypothermic state of 32-34 C helps improve outcomes for SCA patients. There are several theories as to why hypothermia helps preserve ischemic brain tissue, including that it slows cellular metabolism and interrupts the normal progress of apoptosis, helping to preserve cells. For these reasons, it has been hypothesized these same mechanisms of action may greatly expand door-to-balloon time.
“Cooling has two benefits, it slows the metabolism to a crawl so the organs hibernate so you can stop the injury from happening. The second way it helps is it prevents reperfusion injury,” said Christopher White, M.D., FSCAI, FACC, FAHA, FESC, medical director of the John Ochsner Heart and Vascular Institute, Ochsner Medical Center in New Orleans.
Expanding Door-to-Balloon Time
In the United States, hypothermia devices are indicated for restoration of spontaneous circulation after SCA, but White believes data from trials under way will show it should be expanded to acute myocardial infarction (AMI) patients. From experience at Ochsner, about 70 percent of SCA patients suffer an AMI and of the survivors arriving at the hospital, many go on to percutaneous coronary intervention (PCI), White said. Many out-of-hospital SCAs have ECGs performed that do not show STEMI, but he said it is often masked because the patient’s heart is not functioning properly due to the arrest.
New, streamlined American Heart Association/American College of Cardiology guidelines to treat STEMI patients were released in December 2012. These include recommendations for use of hypothermia, stating hypothermia in cardiac arrest patients may help reduce brain injury in these patients and that cooling protocols should be activated before or at the same time of cardiac catheterization.
“Cooling patients does take time, but once you get to therapeutic temperature, door-to-balloon time does not matter anymore because the body is in stasis,” White said.
White said this true in animal experiments, but now clinical trial data is being collected to see if it is also true in human patients. There are several trials in progress to answer this question.
New Clinical Data
One of the key trials is CHILL-MI, which completed enrollment in April 2013 and was included as a late-breaking clinical trial at Transcatheter Cardiovascular Therapeutics (TCT) 2013. The trial evaluated the safety and effectiveness of the Philips InnerCool RTx system in 120 STEMI patients enrolled at nine European centers. Data show that rapidly cooling prior to restoring blood flow is safe, but the trial’s primary endpoint of reducing infarct size was not significantly reduced.
The primary endpoint of the study is reduction of myocardial infarct size as a percentage of myocardium at-risk, assessed by cardiac magnetic resonance imaging (MRI) at four days. Previous research has shown that hypothermia can reduce infarct size in patients whose body temperature reaches ?35 C prior to reperfusion. The previously published Rapid MI-ICE study, which utilized the InnerCool system, showed a 38 percent reduction in infarct size in STEMI patients.
“Hypothermia using intravenous cold saline and an endovascular venous cooling catheter was safe, feasible and reduced temperature to 34.7 C before reperfusion, with a delay of reperfusion of nine minutes,” said David Erlinge, M.D., professor, department of cardiology, Lund University, Skane University Hospital, Sweden, and lead investigator of the study. “Although reduction in infarct size in relation to area at risk did not reach significance, we found a significant reduction in the incidence of clinical heart failure. Furthermore, we observed a large reduction in infarct size in the anterior patient population, but this needs to be validated in a prospective study.”
In the hypothermia group, cooling was initiated before PCI (achieving the target temperature took about 42 minutes) and continued for one hour after reperfusion with a target temperature of 33 C followed by spontaneous rewarming.
Due to the time required for cooling, door-to-balloon time was impacted by an average of nine minutes in the hypothermia group. The primary endpoint, infarct size in relation to the area at risk, was non-significantly reduced by 13 percent (p = 0.15) with similar myocardium at risk between both groups. Although the trial missed its primary endpoint, exploratory analysis indicates an effect in the early anterior patient population with a 33 percent reduction of infarct size.
At 45 ± 15 days, there was no mortality. However, the incidence of clinical heart failure was 14 percent in the control group and 3 percent in the hypothermia group.
Erlinge said due to the statics involved, if the trial had enrolled 200 patients, it would have met its primary endpoint.
The VELOCITY investigational device exemption (IDE) trial is testing the rapid use of therapeutic hypothermia for AMI patients with the Velomedix Inc. system, which is said to achieve 33 C in less than 15 minutes.
The CHIPAHA Trial is slated to begin to test the efficacy of Life Recovery Systems’ ThermoSuit System to cool STEMI patients prior to primary percutaneous coronary intervention (PCI).
The SHOCK-COOL Trial being conducted by the University of Leipzig, Germany, is investigating whether intravascular-induced hypothermia in addition to PCI in acute myocardial infarction complicated by cardiogenic shock improves cardiac power index after 24 hours.
Standard of Care for SCA
“With hypothermia for cardiac arrest, there is very strong evidence for its use. It is a tremendously beneficial treatment for sudden cardiac arrest,” said Marko Noc, M.D., Ph.D., Centre for Intensive Internal Medicine, University Medical Center Ljubljana, Ljubljana, Slovenia, and an expert in therapeutic hypothermia.
He said data from several trials has shown cooling patients helps preserve the brain. Two of the key trials were published in 2002, and shortly after their publication his hospital adopted the use of hypothermia for SCA patients who were comatose following resuscitation. They found the treatment so effective, that it went from being used in a small number of patients to now being a standard of care for 100 percent of these patients.
“It had a significant impact on our survival and neurological outcomes,” Noc said. “For comatose survivors of sudden cardiac arrest, hypothermia is the standard of care, and if physicians don’t use this, they are not properly caring for their patients.”
The University Medical Center Ljubljana serves as the primary acute cardiac care center for all emergency cases in the city of 1.2 million people, so Noc said the center receives a very large volume of patients. From the hospital’s own data registry, in the period between 1995-1997 before the use of hypothermia, Noc said the survival rate of SCA patients was about 25 percent. Of these patients, only 10-15 percent had good neurological outcomes. However, in the years since adopting hypothermia, survival is now about 60 percent and about 50 percent of these survivors had good neurological outcomes.
A similar trend was witnessed at Oscshner, which started use of hypothermia for SCA patients about six years ago. The center gets about four to six SCA cases a month.
“There is nothing that I do for patients that is more beneficial than hypothermia,” White said. “Hypothermia is unbelievable.”
White started running through numerous patient cases from his experience where he believes the patient is still alive and neurologically intact following SCA. In some cases patients’ hearts stopped for more than 30 minutes before they were revived, and they were able to return to their normal lives and jobs almost immediately after their discharge.
“I don’t get the lack of enthusiasm for hypothermia, it’s so easy to do,” White said. “It only takes a few minutes and it is very low tech. It’s really not rocket science.”
Cooling Technology
Technology to cool patients ranges from low simple use of cooled saline and ice packs commonly used pre-hospital by EMS, to in-hospital devices that include cooling pads and suites, or intravascular catheters that circulate cooled saline.
White said the key to hypothermia is how long it takes to get the patient’s temperature into the therapeutic range. For this reason, he is not a supporter of surface cooling systems. For maintaining temperature external systems are fine, but not for emergencies where speed is critical. He said the Medivance Arctic Sun gel pad cooling system takes too long to cool patients, only lowering a patient’s temperature about 1 degree an hour, taking three to four hours to cool a patient.
The ThermoSuit by Life Recovery Systems offers a faster cooling rate of about 3 degrees an hour, with 87 percent of patients achieving target temperatures around 37 minutes.[1] The ThermoSuit cools with direct water on skin contact to draw heat off the body. The water flows over the patient and is returned to the pump.
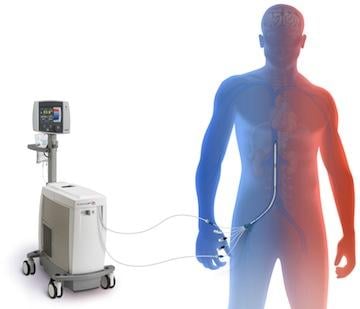
White said intravascular cooling systems take about 30 minutes to achive therapeutic tempturatures. Intravascular catheter-based cooling systems include the Zoll Alsius Coolgard 3000 and the Philips InnerCool. While they are invasive, they can quickly cool patients. The Zoll system uses a balloon and the Philips system uses a metallic catheter. Both connect to a control console that circulates cooled saline through the devices.
Noc also prefers using invasive cooling systems. In addition to taking more time to achieve therapeutic temperatures, he said skin receptors regulate the way a person perceives if they are hot or cold, so patients tend to shiver more with external cooling. With intravascular cooling, Noc said patients are less sensitive to the drop in temperature and can be tricked into feeling warmer by giving them blankets. Ochsner uses the same trick of using warm blankets for conscious patients.
“SCA patients are in a coma, so they are the perfect patients because they don’t shiver,” White said. “Using intravascular systems, they are turned to ice from the inside out.”
Cooling Protocols
The biggest criticisms of the CHILL-MI trial were the protocols used. Cooling was not started by EMS, but only after the patient arrived at the hospital. The average time a patient was cooled prior to PCI was about 42 minutes. A second question raised was the time patients should be cooled after reperfusion is achieved. In CHILL-MI, patients were warmed an hour after PCI, while other centers have protocols to continue cooling for up to day.
“Maybe if you cool a patient longer you may have benefit, but in animal studies we found if you cool after reperfusion you get no benefit,” Erlinge said. “Maybe there would have been more of an effect if we started cooling earlier.”
White said there is not enough clinical evidence to know exactly how long to cool patients to achieve the maximum benefit, so each center may use a different protocol. Both Ochsner and Ljubljana use a similar protocol of cooling patients for 18-24 hours before starting to gradually warm them.
“We do plan with the EMS in New Orleans,” White said. “These guys are the biggest patient advocates and they will find ways to bring us these patients.”
He said EMS uses ice packs and cooled saline to start cooling SCA patients en route to the hospital. This only drops the patients’ temperature a little, so once they arrive invasive cooling is initiated. Noc said his protocol is the same.
References:
1. Daniel Howesa, William Ohleyb, Paul Dorianc. "Rapid induction of therapeutic hypothermia using convective-immersion surface cooling: Safety, efficacy and outcomes." Resuscitation 81 (2010) 388–392.

 April 02, 2019
April 02, 2019 



