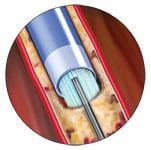
The Spectranetics laser atherectomy system can be used to debulk peripheral vessels without the need for embolic protection.
Atherectomy is a key treatment option in peripheral artery disease (PAD), and several physicians Diagnostic & Invasive Cardiology recently spoke with are fans of the laser over mechanical systems in most PAD cases. They say it is easier to use and eliminates the need for an embolic protection device, but it does have limitations when treating calcified lesions.
Jack Casas, M.D., FACC, head of the endovascular peripheral program at St. Joseph’s Hospital in Parkersburg, W. Va. (formerly of Mercy General in Sacramento, Calif.), has used several atherectomy systems in both coronary and PAD lesions. He likes the Spectranetics laser atherectomy system because it’s versatile, with indications for the coronaries, PAD, vessels below the knee, and for electrophysiology device lead extraction.
No Need for Embolic Protection
“In my experience, distal embolization is rare provided one advances the catheter slowly. It is rare to see a nonreflow complication,” he explained. The laser photo-ablates and completely destroys the plaque. “It basically vaporizes the plaque, turning it into carbon dioxide and water.”
He said the 0.9 mm coronary laser advances about 1 mm per second. It only functions for 10 seconds before shutting down for five seconds. There is no time restriction set by the laser for the peripheral catheters. The system uses a saline flush to provide a shield against damaging red blood cells. The “time out” allows blood to reflow into the area before the physician makes another run.
“It is very easy [to use],” Dr. Casas said. “It is very effective, but you need a little bit of training. Just use some forward pressure and let the laser do its work.”
While it is easy to use, he finds new operators attempt to go too fast. “The biggest thing is to get these guys to go slow,” he said.
Treating PAD
Dr. Casas uses the laser in the majority of his PAD cases, especially in chronic total occlusions (CTOs) in the superficial femoral artery (SFA). “The laser has been very good for me, especially below the knee and in trifurcations,” he said.
The laser system operates over any guide wire, which is not the case with some of the other atherectomy systems. He said another advantage is you can feel the device moving as it cuts through lesions, while mechanical devices generally require an operator to watch the device on fluoroscopy. The laser catheter also tends to follow the lumen very closely, even in tortuous vessels. These factors mean fewer fluoro views of the device, which can reduce patient radiation exposure.
After debulking, Dr. Casas follows up with balloon work. Inflation pressures can be lower in lesions that are debulked, helping lessen the risk of dissection. “The compliance of the vessel is much more friendly,” he said.
However, Dr. Casas added, “By no means is it a stand-alone therapy.”
Atherectomy in Combination With Stents
Stents simply push plaque out of the way instead of removing it. Dr. Casas believes this might be one reason for the high rates of restenosis in stented leg vessels. He uses the laser to debulk the vessel before stenting.
Generally, stents are not used below the knee because it is a hostile environment to the metal tubes. However, Dr. Casas said very high restenosis rates in these vessels has driven him to try new methods to extend the benefit of treatment in some cases. He found paclitaxel-eluting coronary stents work very well at preventing restenosis. Cook Medical is developing a paclitaxel-eluting stent specifically for a PAD indication. However, Dr. Casas said clinical data will have to show clear benefits to justify the significant cost of DES in general practice.
Cutting Through Calcium
Calcified stenoses, especially calcified CTOs, can be weak areas for the laser. “It is OK with calcium, but that is really where the Rotablator works better,” Dr. Casas said.
He has used several mechanical atherectomy devices, including the Pathway Jetstream, Boston Scientific’s Rotablator, CSI’s Diamondback 360, and ev3’s Foxhollow. He likes the concept of Jetstream device, which integrates two different sizes of cutting tips with aspiration in one catheter, but it is limited to work above the knee. For work below the knee, he prefers the Diamondback.


 September 12, 2025
September 12, 2025 









