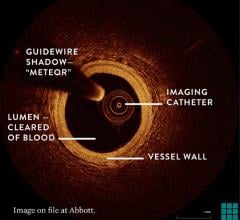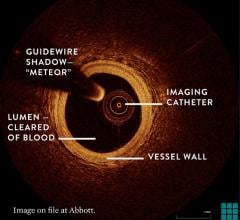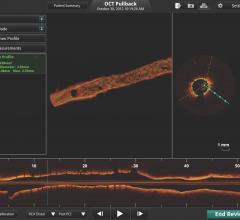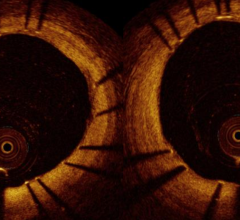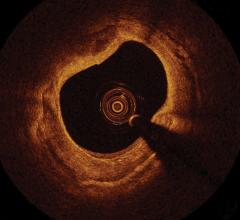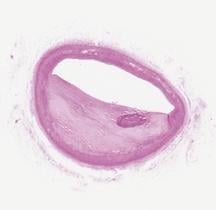
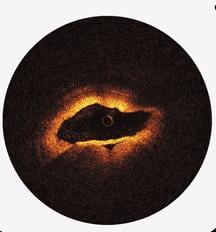
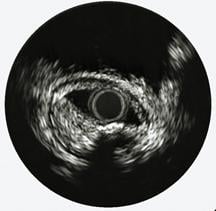
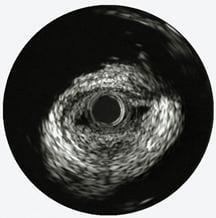
The first three images, courtesy of Professor Takashi Akasaka of Wakayama Medical University and Teruyoshi Kume, M.D., Kawasaki Medical University, shows OCT, IVUS and histological images of the same eccentric fibrous lesion with a calcium deposit.
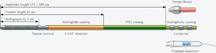
While angiography is the gold standard for vascular imaging in the cath lab, new intravascular imaging modalities may offer innovative ways to get a better picture of stenosis, stent placement and help guide therapy.
Using angiography to see lesions or blockages inside vessels only gives part of the picture. It does not tell the physician how vessels have been remodeled, how much the lesion is impairing blood flow or what the lesion is composed of. Intravascular ultrasound (IVUS) has been around for more than a decade and has become standard equipment in many cath labs. However, other modalities such as optical coherence tomography (OCT), fractional flow reserve (FFR), intravascular MRI and infrared spectrometry are also gaining ground as tools to see what angiography alone cannot.
Intravascular ultrasound
IVUS is the only FDA market-approved intravascular modality in the U.S. It shows the intima, the lumen and areas of plaque, which can all be measured to get an accurate percentage of vessel blockage, measurement of lesion length and help determine the size of stents needed for PCI. IVUS can also be used for post procedural run throughs and follow ups to check if a stent has been fully expanded, to detect stent fracture and to check for and measure restenosis.
IVUS only provides a cross sectional view of a vessel, but development is currently underway by Volcano Corp. to make a forward-looking IVUS system. One of the system’s developers, David O. Williams, M.D., director of the cardiovascular lab and interventional cardiology, Rhode Island Hospital, and professor of medicine at Brown University, explained the system at TCT 2008. He said it will be useful in treating chronic total occlusions (CTOs). He said Volcano may merge the system with a radiofrequency ablation catheter for an intravascular image-guided CTO treatment, where the IVUS could help prevent perforations.
IVUS’ grainy, black and white images are sometimes hard to interpret, which has given rise to new technical innovations. Ik-Kyung Jang, M.D., Ph.D., Massachusetts General Hospital, assistant professor of medicine at Harvard School of Medicine, said IVUS has a resolution of 65 microns and better resolution is needed to visualize certain structures, including identifying components that make up vulnerable plaque.
“If you want to detect fine structural changes, such as vulnerable plaque, IVUS does not have enough resolution,” Dr. Jang said.
The Volcano Corp. introduced IVUS enhanced with virtual histology (VH), which takes the echo returns and creates a color-coded image showing the hardness of materials in a lesion. These can be used to help differentiate four plaque types: fibrous, fibro-fatty, necrotic core and dense calcium. Dr. Jang tested the virtual histology system made by Volcano, but said he was not impressed. He said the central issue is the low resolution of IVUS prevents the images from characterizing plaque on the level needed.
Another limitation on VH-IVUS is that it frequently detects and displays metal stents as calcium or as a necrotic core. A study published in the November 2008 issue of the American Journal of Cardiology (conducted by the Cardiovascular Research Institute/MedStar Research Institute, at Washington Hospital Center in Washington, D.C. and the Cardiovascular Research Foundation) used VH-IVUS in 27 patients with stents. Researchers found 80 percent of struts appeared white and were misclassified as calcium surrounded by red, misclassified as necrotic core; 2 percent appeared just white; and 17 were not detectable. Researchers said it is important not to over interpret VH-IVUS studies of implanted stents when this appearance is observed at follow-up.
Dr. Jang said Japanese researchers developed integrated backscatter IVUS (IB-IVUS), which uses algorithms to distinguish different plaque components and construct a color-coded map similar to VH-IVUS. The map shows thrombus, lipid core, fibrous tissue, mixed lesions and calcification.
Dr. Jang said increasing IVUS resolution beyond its current level might not be possible because higher frequencies cause more echo speckle distortion from blood.
Optical coherence tomography
Dr. Jang is a key investigator for LightLab Imaging Inc.’s OCT system, which is described as a high-definition version of IVUS. LightLab has CE Mark and approval in Japan. The company has more than 300 of its M2 and M2x systems in 20 countries and it has been used in more than 10,000 clinical procedures.
“The biggest advantage with OCT is the resolution, which is 10-15 microns,” Dr. Jang said. “If you put IVUS and OCT images next to each other the difference is night and day.”
OCT catheters use a high-power light source and a detector to capture light that reflects (coherent light) from sub-surface features. Light that scatters from hitting tissue and loses its original direction (non-coherent light) is filtered out.
The first generations of OCT systems required vessel occlusion and aspiration of the blood before imaging, because the light wavelengths used are reflected back by red blood cells. Early OCT systems used a slow time domain algorithm, which required the blood to be removed from the vessel. A next generation of OCT uses the much faster frequency domain. Dr. Jang said this latest generation of OCT from LightLab that is in FDA trials uses a 15 cc clear saline or contrast flush to displace red blood cells for about three seconds so imaging can be done without occlusion.
Giulio Guagliumi, M.D., Cardiovascular Department Ospedali Riuniti di Bergamo, Italy, is the principal investigator for the Optical Coherence Tomography for Drug Eluting Stent Safety (ODESSA) study. At TCT 2008, he said OCT complements, but does not replace IVUS. He said the technology offers resolution 10-30 times higher than IVUS and is much better for near field imaging, making it more useful in visualizing smaller lesions. He also said OCT can visualize thrombus more clearly than IVUS. However, Dr. Guagliumi said the limitations of current OCT technology are shallow depth of light penetration (1-2 mm) where as IVUS can see further into surrounding tissue (about 10 mm).
Volcano is developing an OCT system and had its first human procedure in May 2008 in The Netherlands. Volcano says its OCT imaging platform provides high-resolution images in the 10-15 micron range, which will complement the company's existing line of IVUS imaging catheters. Volcano acquired the OCT technology when it purchased CardioSpectra in 2007. In January it acquired Axsun Technologies Inc., a developer of lasers and optical engines used in OCT.
Intravascular MRI
Intravascular imaging can be done using a wire catheter while a patient is inside a magnetic resonance imaging scanner. Dr. Jang said the technology is not really an imaging modality as much as it is a system to detect and characterize plaque makeup.
The Israeli company TopSpin Medical Inc. developed the Cathamaran Intravascular MRI System that uses a 6F compatible MRI probe to show fibrous tissue in blue and lipids in yellow. The company says the MRI components are miniaturized into the tip of the catheter so a large MRI system is not needed.
TopSpin has submitted a 510(k) application with the FDA, and received CE Mark in August 2007. In January 2008, TopSpin announced its first commercial installation at ZOL Hospital, Genk, Belgium.
Researchers at the Hamburg University of Technology, Institute of MicroSystems Technology, in Germany, said in December they developed an optical transmission system for active intravascular MRI devices. The system avoids the use of long conducting cables, which due to resonance can cause tissue heating. An optical transmitter sends the MR signal via an optical fiber.
Spectrometry
The LipiScan spectrometry imaging system from InfraReDx uses a catheter that beams near-infrared light into the plaque and reflected wavelengths of light are used to determine the chemical composition of plaque. The system produces a chemograph to show the makeup of the plaque being imaged, but is not a true imaging modality.
“I don’t think spectrometry will survive as a stand-alone modality, it needs to be combined with IVUS or OCT,” Dr. Jang said.
For more information:
www.axsun.com
www.bostonscientific.com
www.lightlabimaging.com
www.radi.se
www.volcanocorp.com




 August 29, 2023
August 29, 2023