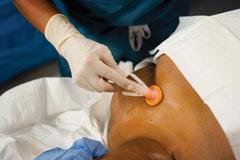
The 2002 CDC guidelines on preventing intravascular device-related infections recommend using a chlorhexidine gluconate (CHG) solution. CareFusion offers ChloraPrep applicators with a solution of 2 percent CHG/70 percent isopropyl alcohol.
Editor’s note: Jerome Granato, M.D., is the medical director of the coronary care unit at Allegheny General Hospital in Pittsburgh, Pa. His facility started looking at ways to cut its nosocomial infection rates several years ago. This issue has a broader importance today, with CMS now cutting reimbursements for hospital-acquired infections and other “never events.”
Central venous catheters (CVCs) are frequently employed for both diagnostic and therapeutic purposes, but their use is associated with the risk of complications. The most significant complication is catheter-related bloodstream infections (CRBSIs). Until recently, most clinicians considered these infections to be an unavoidable and an accepted complication of complex medical care. There is now growing scientific information and clinical evidence that clearly suggests otherwise. Many CRBSIs are indeed preventable and pose an unacceptable risk for patients, and place an undo burden on the healthcare industry.
Approximately 250,000 intravascular-related bloodstream infections (BSIs) occur in U.S. hospitals each year,(1) with just CVC-related BSIs resulting in a mortality rate of 20 percent.(2) The estimated annual cost of caring for these patients ranges from $296 million to $2.3 billion.(3)
Allegheny General Hospital in Pittsburgh, Pa., asked what it could do in 2003 when it began a journey to eliminate CRBSIs in the coronary care unit. We knew eliminating these infections would not only benefit our patients, but it would be less expensive.
The first step in revising our infection prevention efforts was to evaluate our central line insertion practices, identify potential sources of infection and to design and adopt corrective measures. We assembled a multidisciplinary team to observe the insertion and care of these lines over several weeks. The observers were amazed to find marked variations in the way these lines were inserted and cared for. We felt by reducing the variation in insertion and maintenance techniques, we would be able to reduce the opportunity of introducing infection. The team made recommendations to standardize the process of CVC insertion and care for all doctors and nurses. These included shifting the standard site of central line insertion from the femoral vein to the subclavian or jugular vein, eliminating the practice of rewiring existing lines and establishing a protocol of removing and replacing central lines of new patients coming into the hospital.
In emphasizing upper torso central line insertion, we assumed most physicians were familiar and comfortable with this technique. To our surprise, we found a wide diversity of knowledge and experience among physicians with respect to the fundamentals of aseptic technique and central line insertion. Unfortunately, medical and nursing schools often do not include formal training in these techniques. This “ad hoc” approach to central line insertion resulted in the type of variability we wanted to avoid, and facilitated infections.
We implemented an infection prevention curriculum and central line insertion training program for all residents, fellows and nurses. The program is still in use today and spans the entire process, including basic anatomy, appropriate sterile technique and skin preparation, line insertion methods, appropriate care for catheters, and the recognition and management of catheter-related complications. We are able to administer this program to large numbers of people by placing it online on the hospital intranet. Everyone who is involved in CVC insertion is annually required to confirm their understanding of the principles by passing a written test. Physicians are further required to demonstrate their competency with the insertion techniques during a 30-minute simulation session with a mannequin and a proctor.
Standardizing the process and the training has proven to be exceptionally successful. Prior to implementation, the institution had an average rate of 5.1 infections per 1,000 line-days. Now, we have sustained a zero percent infection rate in the coronary care unit and trauma care unit for periods up to 16 consecutive months. Over the past five years, the institutional rate central line infection rate has fallen annually to a current rate of 0.2 infections per 1,000 line-days. In contradiction to our previous notion, central line infections are the exception and not an acceptable complication of complex
medical care.
The central line training module and infection prevention protocols we developed are based on observations and recommendations made by front line workers who actively participate in central line insertion, scientific evidence published by the CDC(4) and the central line bundle approach developed by the Institute for Healthcare Improvement.(5) Here are some practices to consider when implementing a central line infection prevention program:
Sterile, Aseptic Technique
CRBSIs are caused by naturally occurring microorganisms on the patient’s skin and on the hands of healthcare workers. They enter the body through the catheter insertion site either at the time of catheter placement, or after several days following catheter manipulation or dressing changes. For this reason, strict aseptic technique and scrupulous skin preparation must be employed. Everyone who participates in the central line insertion process or daily care must wash their hands with soap and water or an alcohol cleanser, even if gloves are worn. Maximum barrier precautions must be employed at all times. This includes a full gown, face mask, hair covering and sterile gloves for the operators, as well as face mask and hair covering for all persons in the room. The patient should also be fully covered with a fenestrated sterile drape.
Properly prepping the patient’s skin is a key component of infection prevention. The 2002 CDC guidelines on preventing intravascular device-related infections include the recommendation to use a 2 percent chlorhexidine gluconate (CHG) base preparation for antisepsis.(4) I recommend a 2 percent CHG/70 percent isopropyl alcohol (IPA) solution such as ChloraPrep. The immediate action from the IPA helps to sterilize the skin and ensure that bacteria are not introduced into the insertion site. The residual activity from CHG reduces the risk of bacterial regrowth that could potentially migrate into the insertion site and colonize on the biofilm that frequently forms around the catheter. In addition, this particular skin prepping product comes with an applicator, which minimizes direct hand-to-patient contact, reducing cross contamination.(6)
For patients with a heavy hair burden, some removal may be necessary. According to the CDC and the Surgical Care Improvement Project (SCIP), insertion site shaving with a razor is associated with a higher risk of nicks and dermal abrasions that can increase the risk of infection. Electric surgical clippers should be used that offer wet shaving. A clipper designed for wet hair removal also allows for full submersion in disinfectants for thorough cleaning in between patient procedures. Also, look for a clipper that is designed to go through hair in a single pass for minimum skin disruption.
Maintenance Care
The healthcare team should inspect the insertion site daily for any sign of infection. Clinicians should review the need for the line and remove it promptly once it is no longer necessary. All CVCs require regular maintenance and care, therefore, it is important to know which type of catheter your facility is using so you will know the level of care that is required. With all maintenance procedures, aseptic technique should be used. Some general maintenance guidelines include:
• Injection Caps: Manufacturer instructions will vary, but typically caps should be changed weekly. To prevent contamination of the catheter, the top of the cap should be disinfected with alcohol each time a syringe or IV tubing is attached to a one-piece injection cap.
• Flushing: There is no established flushing standard for CVCs, however, each facility should establish a protocol, as flushing helps decrease the risk of occlusion and assists in maintaining patency. In our facility, we flush lines with non-heparinized saline, once every eight-hour shift.
• Dressing Changes: Dressings should be changed once a week if using a transparent semipermeable membrane, or twice a week for all other dressings. Transparent dressings are favorable, as they permit observation of the insertion site without disruption of the protective barrier. Dressing should also be changed if they become soiled, loose, and damp or if the patient complains of discomfort.
Infection Surveillance
It is imperative that an infection surveillance program includes real-time reporting and adaptation. We established a weekly infection review called a “bug meeting.” All members of the healthcare team including physicians, nurses, infection “preventionists” and even housekeeping participate.
The elimination of CRBSIs is everyone’s responsibility. Any infections are discussed in an open but nonthreatening manner, to determine what broke down and how the process can be improved moving forward. The elimination of CRBSIs is a never-ending journey. It is not a destination, but rather requires a continuous commitment from all members of the healthcare team as well as hospital administration.
References:
1. Crnich CJ, Maki DG. “The Promise of Novel Technology for the Prevention of Intravascular Device–Related Bloodstream Infection. I. Pathogenesis and Short-Term Devices.” Healthcare Epidemiology. CID. May 2002; 34: 1232-1242.
2. Institute for Healthcare Improvement. “Getting Started Kit: Prevent Central Line Infections. How-to Guide.” 2006. Available at www.ihi.org/nr/rdonlyres/0ad706aa-0e76-457b-a4b0-78c31a5172d8/0/centrallineinfectionshowtoguide.doc. Accessed Sept. 3, 2009.
3. “CDC Morbidity and Mortality Weekly Report.” Aug. 9, 2002. Vol. 51/No. RR-10.
4. Adams D, Quayum M, Worthington T, Lambert P, Elliott T. “Evaluation of a 2% chlorhexidine gluconate in 70% isopropyl alcohol skin disinfectant.” Journal of Hospital Infection. 2005; 61: 287-290.
5. Institute for Healthcare Improvement. “Getting Implement the Central Line Bundle.” Available at www.ihi.org/IHI/Topics/CriticalCare/IntensiveCare/Changes/ImplementtheCentralLineBundle.htm. Accessed Sept. 17, 2009.
6. Denton GW. Chlorhexidine. In: “Block SS. Disinfection, Sterilization, and Preservation.” 5th edition. Philadelphia, Pa: Lippincott Williams & Wilkins; 2001:321-336.

