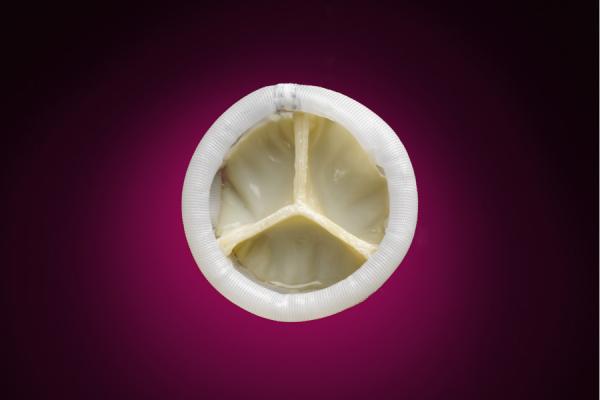
September 11, 2013 — Direct Flow Medical Inc. announced the first patient enrollment in the U.S. SALUS clinical trial. The trial will study the Direct Flow Medical Transcatheter Aortic Heart Valve System, which encompasses a distinctive transcatheter aortic heart valve with a metal-free frame and flexible, low-profile delivery system designed to eliminate aortic regurgitation. The device is intended to improve the long-term survivability of aortic stenosis patients by resolving the clinical issues associated with current commercial valves.
The SALUS Trial is a non-randomized, multi-center, core lab-adjudicated, investigational device exemption (IDE) trial of 30 patients being conducted at six U.S. clinical sites. Principal investigators for the SALUS Trial are Murat Tuzcu, M.D., vice chairman of the department of cardiology, Cleveland Clinic, and Patrick McCarthy, M.D., director of the Bluhm Cardiovascular Institute and chief of cardiac surgery, Northwestern Memorial Hospital.
William O’Neill, M.D., medical director of the Center for Structural Heart Disease at Henry Ford Hospital performed the first Direct Flow Medical case in the United States.
"The team at Henry Ford Hospital is pleased to be the first in the U.S. to implant the Direct Flow Medical heart valve," said O'Neill. "A device that is repositionable and able to pass through smaller diameter blood vessels is an important advance in the next generation of transcatheter aortic valve replacement (TAVR) systems. This could help patients who have not been good candidates for earlier TAVR devices."
The Direct Flow Medical Transcatheter Aortic Valve System is designed to achieve optimal outcomes by enabling in-situ hemodynamic assessment after the valve is fully deployed in the native valve annulus, as well as repositioning with full distal, proximal and planar control, or retrieval, if required. The system avoids rapid pacing of the heart during deployment, and does not require post-dilatation following placement, minimizing the risk of hemodynamic stress for patients.
The Direct Flow Medical System received the CE mark in January 2013 and is currently available commercially in Europe.
For more information: www.directflowmedical.com


 December 24, 2025
December 24, 2025