January 15, 2014 — A new
magnetic resonance imaging (MRI) method to map creatine at higher resolutions in the heart may help clinicians and scientists find abnormalities and disorders earlier than traditional diagnostic methods, suggest researchers at the Perelman School of Medicine at the University of Pennsylvania in a new
study published online in
Nature Medicine. The preclinical findings show an advantage over less sensitive tests and point to a safer and more cost-effective approach than those with radioactive or contrasting agents.
Creatine is a naturally occurring metabolite that helps supply energy to all cells through creatine kinase reaction, including those involved in contraction of the heart. When heart tissue becomes damaged from a loss of blood supply, even in the very early stages, creatine levels drop. Researchers exploited this process in a large animal model with a method known as chemical exchange saturation transfer (CEST), which measures specific molecules in the body, to track the creatine on a regional basis.
The team, led by Ravinder Reddy, Ph.D., professor of radiology and director of the Center for Magnetic Resonance and Optical Imaging, Penn Medicine, found that imaging creatine through CEST MRI provides higher resolution compared to standard magnetic resonance spectroscopy (MRS), a commonly used technique for measuring creatine. However, its poor resolution makes it difficult to determine exactly which areas of the heart have been compromised.
“Measuring creatine with CEST is a promising technique that has the potential to improve clinical decision making while treating patients with heart disorders and even other diseases, as well as spotting problems sooner,” said Reddy. “Beyond the sensitivity benefits and its advantage over MRS, CEST doesn’t require radioactive or contrast agents used in MRI, which can have adverse effects on patients, particularly those with kidney disease, and add to costs.”
MRI-based stress tests are also used to identify dead heart tissue — which is the warning sign of future problems such as coronary artery disease (CAD) — but its reach is limited. MRI is often coupled with contrast agents to help light up problem areas, but it is often not sensitive enough to find ischemic, but not yet infarcted, regions with deranged metabolism, according to Reddy.
“After a heart attack, different regions of the heart are damaged at different rates,” said Robert Gorman, M.D., professor of surgery and director of cardiac surgical research, Penn Medicine. “This new technique will allow us to very precisely study regional changes that occur in the heart after heart attacks, enabling us to identify and treat patients at risk for developing heart failure before symptoms develop.”
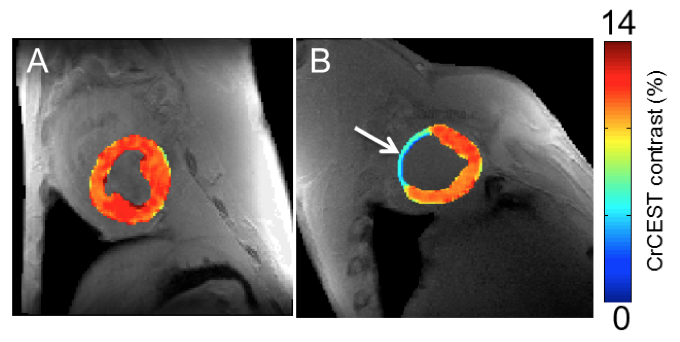
To demonstrate CEST’s ability to detect heart disease, the researchers applied the creatine CEST method in an MRI scanner in healthy and infarcted myocardium in large animals. In the process, the nuclear magnetization of amine (NH2) creatine protons is saturated by a radiofrequency pulse from the MRI. After the exchange with water, the degree of saturation is observed as the water signal drops, and thus the concentration of creatine becomes apparent.
The team showed that the creatine CEST method can map changes in creatine levels and pinpoint infarcted areas in heart muscle tissue, just as MRS methods can. However, they found CEST has two orders of magnitude higher sensitivity than MRS. That advantage could help spot smaller damaged areas in the heart missed by traditional methods, the authors said.
In addition, the team used CEST to map increases in creatine over time by imaging human subjects as they flexed their calves while inside an MRI scanner to demonstrate the technology’s ability to correctly track the molecule.
The method can also be used to investigate alterations in normal heart function that are seen in many other types of non-ischemic heart disease, such as abnormal cardiac hypertrophy as well as disorders in the brain, according to Reddy.
“Though at much lower levels than in the heart, creatine levels change in the brain when abnormalities arise,” said Reddy. “Given the heightened resolution of this technique, this presents an opportunity for studying brain disorders with deranged creatine metabolism without the use of contrast agents as well.”
CEST has been used to image tissue pH, map proteins and specific gene expression, but this is the first time to the authors’ knowledge it has been used to study heart tissue.
"The ability to visualize heart muscle viability at high resolution without radiation exposure or the injection of a contrast agent is a significant advancement,” said Christina Liu, Ph.D., who oversees funding for molecular imaging research by the National Institute of Biomedical Imaging and Bioengineering (NIBIB), part of the National Institutes of Health. “It could allow doctors to detect small areas of damaged heart tissue early in the course of a disease when treatments are most likely to be effective.”
Penn Medicine co-authors of the study include Mohammad Haris, Anup Singh, Kejia Cai, Feliks Kogan, Jeremy McGarvey, Catherine DeBrosse, Gerald Zsido, Walter RT Witschey, Kevin Koomalsingh, James Pilla, Julio Chirinos, Victor Ferrari, Joseph Gorman and Hari Hariharan.
The study was funded with grants from the NIBIB (P41EB015893S1, P41EB015893, and R21DA0332256-01) and a pilot grant from the Translational Biomedical Imaging Center of the Institute for the Translational Medicine and Therapeutics of the University of Pennsylvania.
For more information: www.med.upenn.edu, www.nature.com