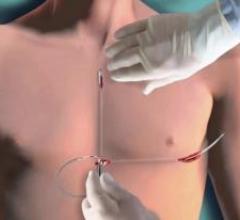
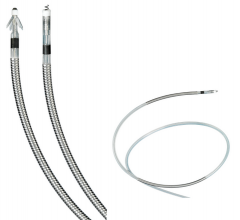
December 18, 2012 — Zoll Medical Corp. announced that its new U.S. Food and Drug Administration (FDA)-cleared OneStep ...
Leads Implantable Devices
News and new technology innovations for leads implantable devices can be found on this channel.
October 29, 2012 — Boston Scientific Corporation has received regulatory approval to market the Reliance 4-Front lead ...
October 8, 2012 — The U.S. Food and Drug Administration (FDA) has granted Boston Scientific Corp. regulatory approval ...
August 3, 2012 — St. Jude Medical recently announced initial findings from the Riata lead evaluation study. The study’s ...
June 25, 2012 — St. Jude Medical Inc. today commented on the Durata lead failure reported in the MAUDE database report ...
June 21, 2012 — A class action suit has been filed against St. Jude Medical Inc. on behalf of purchasers of the company ...
June 8, 2012 — Boston Scientific Corp. has closed its acquisition of Cameron Health, Inc. of San Clemente, Calif., and ...
Data was presented during the Heart Rhythm Society (HRS) 2012 scientific sessions that showed the Riata ICD lead had a ...
Heart Rhythm Society (HRS) President-Elect Hugh Calkins, M.D., FACC, director, cardiac arrhythmia services and EP lab ...
Pivitol trial data regarding the safety and efficacy of the Cameron Health Subcutaneous Implantable Cardioverter ...
May 14, 2012 — Sorin Group at the Heart Rhythm Society (HRS) scientific sessions announced U.S. Food and Drug ...
May 11, 2012 — The Sorin Group is launching new implantable devices with a proprietary algorithm system as well as three ...
May 2, 2012 — The Spectranetics Corp. announced U.S. Food and Drug Administration (FDA) approval of the new advanced ...
May 2, 2012 — St. Jude Medical Inc. yesterday released its biannual product performance report (PPR) on its website, www ...


 December 18, 2012
December 18, 2012