May 21, 2008 - Physicians can detect and diagnose clinically actionable events earlier in pacemaker patients who utilize ...
Pacemakers
This channel includes news and new technology innovations for pacemakers used to treat bradycardia.
May 14, 2008 - St. Jude Medical received both FDA 510(k) clearance and European CE Mark approvals of the IsoFlex ...
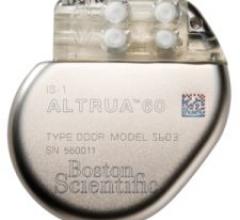
Boston Scientific's ALTRUA family of pacemakers is the first Boston Scientific-branded pacemaker to treat bradycardia.
...
May 9, 2008 - The FDA cleared Boston Scientific's ALTRUA family of pacemakers, a day after the European approval of ...
April 22, 2008 - The cardiac pacemakers market is expected to reach $3.7 billion by 2010, according to the report ...

Choosing the right device to regulate or jump-start the heart can be a challenge. Not only does a patient’s ...
March 3, 2008 - Boston Scientific Corp. received CE Mark approval of its ACUITY Spiral left ventricular lead for ...
March 3, 2008 – The FDA granted 510(k) clearance for the AIGISRX CRMD Anti-Bacterial Envelope by Tyrxpharma, a ...

When cardiologists send their high-risk patients home, they may be protected with a host of tools, including ...
January 28, 2008 – Medtronic Inc. has launched a clinical trial to confirm the safety and efficacy of the Medtronic ...
January 22, 2008 - The International Board of Heart Rhythm Examiners (IBHRE) issued the first standardized cardiac ...
November 13, 2007- There is significant underutilization of many guideline-indicated life-saving medical and ...
September 19, 2007 - Biotronik announced that it has been awarded a full-service cardiac rhythm management (CRM) ...
September 6, 2007 – The New England Journal of Medicine published the first study to demonstrate a clear superiority of ...
August 29, 2007 - The European Society of Cardiology has released new ESC Guidelines for Cardiac Pacing and Cardiac ...

 May 20, 2008
May 20, 2008