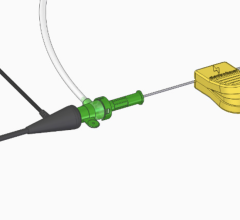
The success or failure of coronary intervention starts with a winning combination of guiding catheter and coronary guidewire, which gives the operator the optimal support and stability to accomplish the interventional goals. The Terumo Runthrough NS coronary guidewire has some unique properties, which allows access to a broad range of complex anatomies. The guidewire is a hybrid wire combining the support of stainless steel and the memory and flexibility of nitinol. The distal 40 cm of the wire is a one-piece tapered nitinol core with the distal tip overlayed with platinum (3 cm) and stainless steel (22 cm) coils. The most distal (2 mm) is coated with silicon, which affords greater tactile feel and the remaining approximately 25 cm is hydrophyllic coated. The balance of the nitinol and stainless steel shaft are coated with PTFE to complement the lubricity of the distal end. A primary feature of the Runthrough NS is how the nitinol and stainless steel are fused directly together, which provides exceptional torque transmission, while maintaining guide catheter support. It appears that the Runthrough wire has a distinguishing characteristic that separates it from most traditional wires: Its one-piece nitinol tip maintains its shape after going through the coronary lumen and throughout the procedure. This is a unique advantage over most wires that utilize a stainless shaping ribbon. After testing the performance of the Runthrough, I then used it as my primary wire for all coronary interventions with the exception of obvious chronic total occlusions for four months. In 90 cases performed thus far, I have had only one instance where the Runthrough NS was unsuccessful.
The case presentation below represents a typical example of how this wire performs.
A 72-year-old Caucasian male underwent coronary artery bypass grafting in 1991 for severe ischemic coronary disease and limiting angina. A LIMA was placed to the mid LAD and a vein graft to the diagonal branch of the LAD. A separate saphenous vein was placed to the mid body of a large dominant PDA, which extends to the apex of the left ventricle and supplied the entire apex. The distal right coronary artery was comprised of a moderate to large posterior lateral branch, which was filled in a retrograde manner from this saphenous vein graft.
The patient did extremely well with no major adverse cardiovascular events until September of 2007 when he was presented to his cardiologist with complaints of daily angina, requiring supplemental sublingual nitroglycerine. He was, therefore, placed in the hospital and was referred for coronary angiography followed by revascularization if needed.
The patient did rule out for an acute myocardial infarction by EKG and ischemic markers.
On Sept. 5, 2007, coronary angiography revealed that the left ventricular systolic function was well preserved with an ejection fraction of 50 percent. The LIMA to the LAD and the vein graft to the diagonal branch were widely patent with no obstructions to flow. There was severe disease in the right coronary vein graft.
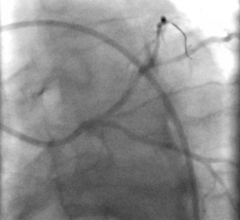
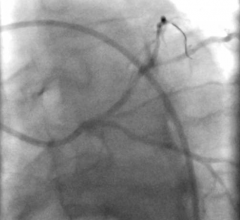
Angiogram of the vein graft to the PDA with primary lesion is noted in Fig. 1.
The challenge of this coronary intervention was to be able to access the PDA antegrade, as well as accessing retrograde back to the AV groove and out to the posterior lateral branch. This would ensure the ability to revascularize the insertion site of the vein graft into the PDA, as well as to revascularize the posterior lateral branch, which had a significant retrograde stenosis, as well.
The patient was anticoagulated with Angiomax. The guiding catheter used was a 7-French short-tipped right guiding catheter. The Runthrough wire was placed in the distal PDA. Intravascular ultrasound revealed severe plaque prior to insertion into the PDA.
The antegrade limb of the PDA was no more than 2.5 mm in diameter by IVUS. A second Terumo wire was then placed into the retrograde limb of the PDA leading back to the posterior lateral branch (Fig 2). This allowed for access to the retrograde limb with ultrasound, which revealed that the retrograde PDA was approximately 3 mm in diameter.
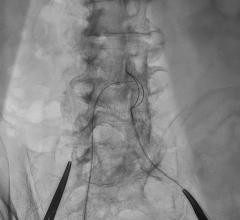
A 3 mm-by-20 mm Taxus stent was placed into the retrograde limb of the PDA and brought into the vein graft. The PDA wire was left in as a safety wire. It was then removed and repositioned through struts of the stent back into the antegrade PDA limb. This was then followed by a 4 mm high-pressure balloon inflation in the vein graft and then followed by kissing balloons with a 2.5 mm balloon in the antegrade limb and a 3 mm balloon in the retrograde limb. Because of the extreme tortuosity going to the posterior lateral branch, balloon angioplasty only was done of this segment. Final results are presented in Fig 3.
The patient did well and there were no identifiable major adverse cardiovascular events.
The performance of the Runthrough NS wire improves efficiencies because of its ability to traverse simple and complex anatomies in an extremely timely manner. The way this wire is engineered also allows for the same wire to be used on different vessels during the same procedure because the wire tip tends to maintain its shape as opposed to other coronary guidewires; or may be reshaped if needed (something that is not routinely possible with wires that have stainless shaping ribbons). This, in effect ,will reduce costs since only one wire is needed. In addition, it may improve patient care because the wire appears to be quite safe in the distal coronary circulation. The wire crosses stent struts into side branches with little difficulty. Overall, “wire time” is less with the Runthrough NS wire.
In summary, the Terumo Runthrough coronary guidewire is an excellent advancement in transcatheter coronary revascularization.
Case Study | March 11, 2008 | Stephen A. Lewis, M.D., FACC, Associated Cardiology of Charleston, PLLC


 June 17, 2024
June 17, 2024