June 20, 2017 — Dutch medical device company Stroke2prevent BV recently announced it has…
June 16, 2017 — The U.S.
June 12, 2017 — Abiomed Inc.
June 12, 2017 — TandemLife began its commercial launch of the TandemLife Priming Tray, a new…
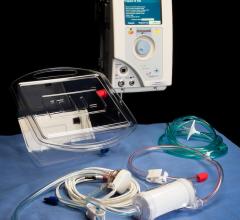
June 1, 2017 — Infab Corp.
MIM Software Inc. recently announced significant updates to its MIM Encore solution for viewing…
The U.S. Food and Drug Administration (FDA) announced that it has granted market clearance to Ra…
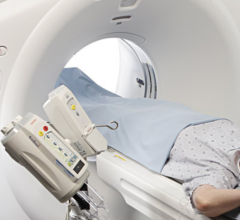
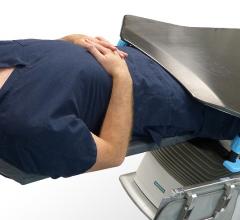
Adept Medical announced the launch of the IR Platform, designed as an over-patient work surface…
May 15, 2017 – The U.S.
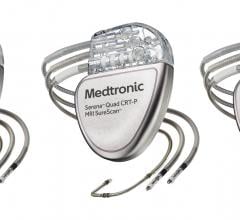
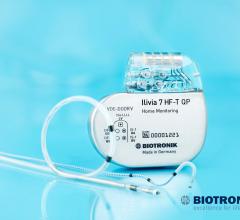
May 11, 2017 — Biotronik announced U.S.
Biotronik announced the availability of the first U.S. Food and Drug Administration (FDA)-…
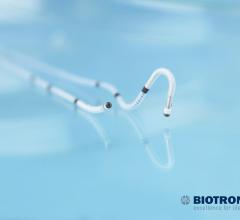
Biotronik announced U.S. Food and Drug Administration (FDA) approval and the launch of Sentus…
May 9, 2017 — The U.S.
May 2, 2017 — The U.S.
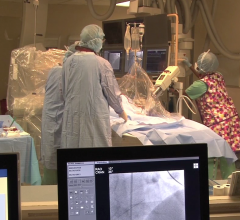
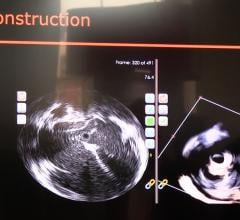
Conavi Medical Inc. announced 510(k) clearance from the U.S. Food and Drug Administration (FDA)…
May 1, 2017 — The U.S.
Physio-Control announced April 19 that the company’s HeartSine samaritan PAD 360P (SAM 360P)…
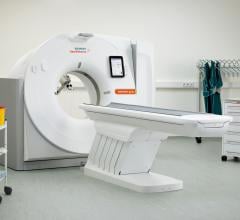
The U.S. Food and Drug Administration (FDA) has cleared the Somatom go. computed tomography (CT…
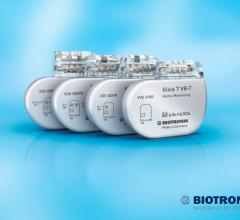
March 23, 2017 — Biotronik announced the European launch of the Edora series, its smallest…
Aegis Medical Innovations Inc. announced that it has received Investigational Device Exemption…


 June 20, 2017
June 20, 2017