Heart Imaging Technologies announced the release of Precession, a cardiac magnetic resonance…
New and updated American College of Radiology (ACR) Appropriateness Criteria now help healthcare…
CompView Medical (CVM) introduced an all-in-one equipment manager, visualization and ergonomic…
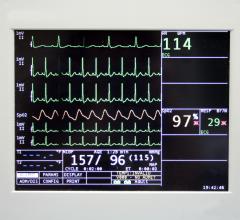
The ScottCare Corp. announced its new novi patch Holter monitor will be available in the United…
Pie Medical Imaging BV announced that it received 510(k) clearance from the U.S. Food and Drug…
Agfa Healthcare announced the commercial launch of its Enterprise Imaging for Cardiology Suite…
Calgary Scientific Inc. announced the release of ResolutionMD 5.0, which offers better access to…
Cerner Corp. and development partner Mortara Instrument have joined together to introduce…
Physicians who use the clinical reference tool, …
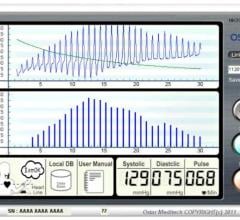
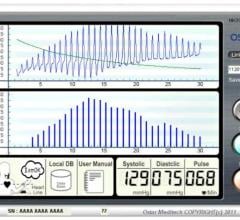
Ostar Healthcare Technology announced the North American launch of their Cellular P201…
Samsung Electronics America introduced the Samsung WS80A with Elite performance package, its…
Ostar Healthcare Technology, a Washington State Based TeleHealth Solutions company, announced…
AliveCor Inc. announced it has received CE Mark clearance for its automated analysis process (…
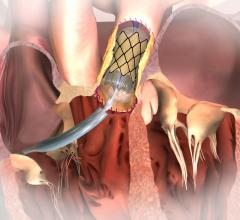
St. Jude Medical Inc. announced U.S. Food and Drug Administration (FDA) approval of the…
Maquet Medical Systems USA announced an agreement to serve as the exclusive U.S. distributor of…
The U.S. Food and Drug Administration (FDA) has…
Kopp Development Inc. released a new entryway system — FerrAlert Halo II Plus.
The U.S. Food and Drug Administration (FDA) has granted a humanitarian device exemption for the…
Ambry Genetics announces the launch of cardiovascular genetic testing panels for inherited…
Daiichi Sankyo Co. Ltd. announced that the U.S. Food and Drug Administration (FDA) has approved…

 March 03, 2015
March 03, 2015