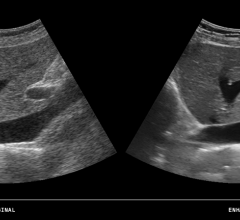
Carestream has obtained U.S. Food and Drug Administration (FDA) clearance for diagnostic reading…
At this year’s scientific assembly and annual meeting of the Radiological Society of North…
Rijuven announced the launch of CardioSleeve for Pediatrics, the first U.S. Food and Drug…
Philips announced that Lumify, a smart device ultrasound solution, is available for purchase by…
iRhythm Technologies Inc. announced the launch of new patient engagement tools to enhance the…
Fujifilm SonoSite Inc. announced it has received 510(k) clearance from the U.S. Food and Drug…
Kopp Development Inc. announced the release of the new accessory for their latest magnetic…
Access Scientific LLC announced validation of the Powerwand midline IV catheter for power-…
Stereotaxis Inc. announced the worldwide launch of Respiratory Compensation, a new software…
The U.S. Food and Drug Administration (FDA) granted accelerated approval to Praxbind (…
October 15, 2015 — U.S.
Kopp Development Inc. has released a new entryway system for magnetic resonance imaging (MRI)…
Shimadzu Medical Systems USA announced the availability of the MIX package for the Trinias…
Personal MedSystems introduced the first and only 22-lead mobile electrocardiogram (ECG) system…
Invitae Corp., a genetic information company, announced the expansion of its cardiology offering…
October 5, 2015 — The U.S.
AtriCure Inc. launched the cryoFORM cryoablation probe, which offers increased probe flexibility…
AstraZeneca announced that Brilinta (ticagrelor) 60-mg tablets are now available in U.S.…
Galt Medical has partnered with Health Line International Corp. and launched the Nexus CT…
AstraZeneca announced that the U.S. Food and Drug Administration (FDA) has approved Brilinta (…

 November 20, 2015
November 20, 2015