August 20, 2014 — Vital Images Inc. received 510(k) clearance from the U.S. Food and Drug…
Teleflex Inc.’s subsidiary Hotspur Technologies Inc. received U.S. Food and Drug Administration…
August 11, 2014 — Medtronic announced it has received U.S. Food and Drug Administration (FDA)…
August 11, 2014 — NeoCoil LLC announced it has received 510(k) clearance from the U.S. Food and…
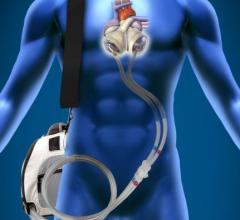
SynCardia Systems Inc. received approval in July from the U.S. Federal Drug Administration (FDA…
OSTAR Healthcare Technology, a Washington State-based Medical Device and Healthcare IT company,…
August 4, 2014 — Ostar Healthcare Technology, a Washington-based medical device and healthcare…
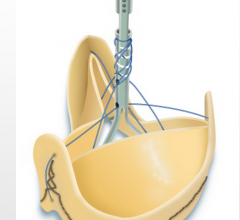
Sorin Group received U.S. Food and Drug Administration (FDA) approval for the Solo Smart Aortic…
August 1, 2014 — MedXT, a cloud platform provider for medical imaging, launched a cost-effective…
July 30, 2014 — Covidien announced the commercial launch of its next-generation Trellis…
CorMatrix Cardiovascular announced that it has received U.S. Food and Drug Administration (FDA)…
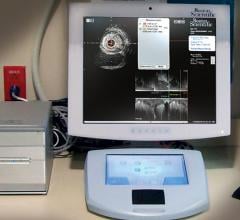
July 24, 2014 — Boston Scientific has initiated full commercial launch of its new Polaris…
July 22, 2014 — Boston Scientific announced it received U.S. Food and Drug Administration (FDA)…
eMix has released Version 2.0 of its cloud-based medical-image exchange solution — including a…
July 18, 2014 — Cook Medical customers in the United States will again have access to its…
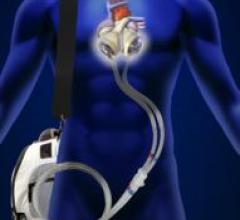
July 18, 2014 — The Freedom portable driver has received U.S. Food and Drug Administration (FDA…
Objective Medical Systems LLC announced that its Electronic Health Record (EHR) system has…
The ScottCare Corp. announced the newest version of its multi-parameter telemetry solution,…
July 11, 2014 — Cordis Corp. announced the launch of its Saber PTA (percutaneous transluminal…
July 11, 2014 — Toshiba America Medical Systems introduced its newest M-Power software version…

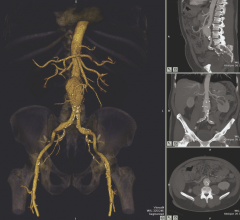
 August 20, 2014
August 20, 2014