June 21, 2012 — TriReme Medical Inc. (TMI) announced today that it has received U.S. Food and…
June 21, 2012 — Surefire Medical Inc. announced that it has received 510(k) U.S. Food and Drug…
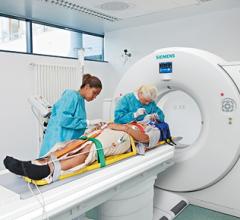
June 20, 2012 – Siemens Healthcare announced the Somatom Definition Edge single-source computed…
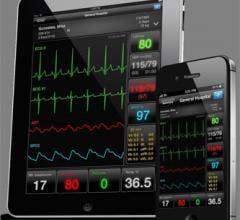
June 18, 2012 — AirStrip Technologies Inc. has expanded its platform to deliver information…
June 18, 2012 — St. Jude Medical Inc. announced it has received U.S. Food and Drug…
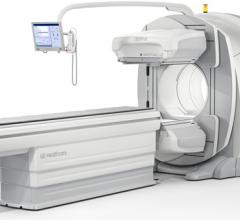
GE Healthcare introduced the latest addition to its Nuclear Medicine 600 series with a new…
June 15, 2012 — Cardiosolutions Inc. announced it has received 510(k) clearance from the U.S.…
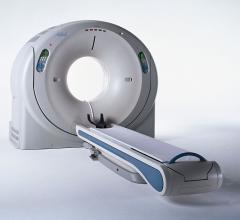
June 15, 2012 — To enable hospitals to keep pace with the latest technological advances and…
June 14, 2012 — Fujifilm Medical Systems USA Inc. announced the availability of Synapse Mobility…
Best Cyclotron Systems is unveiling five new cyclotrons for isotope research and production at…
June 8, 2012 — Bayer HealthCare announced the formation of a strategic partnership with…
June 11, 2012 — Critical Diagnostics, a U.S.-based biomarker company focused on cardiovascular…
June 11, 2012 — Calgary Scientific Inc. has received clearance from the U.S. Food and Drug…
June 8, 2012 — Bayer Radiology and Interventional has announced the introduction of the Certegra…
June 8, 2012 — Medtronic Inc. announced the U.S. launch of the Endurant II AAA Stent Graft…
Hansen Medical Inc. received 510(k) market clearance from the U.S. Food and Drug Administration…
June 4, 2012 — Boston Scientific Corp. has received U.S. Food and Drug Administration (FDA)…
June 1, 2012 — Civco Medical Solutions is introducing a newly redesigned storage rack and…
May 30, 2012— Boston Scientific Corp. announces the U.S. Food and Drug Administration has…
May 23, 2012 - TriReme Medical Inc. announced approval by the U.S. Food and Drug Administration…

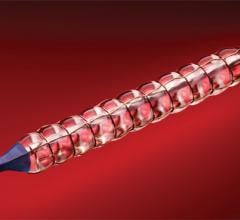
 June 21, 2012
June 21, 2012