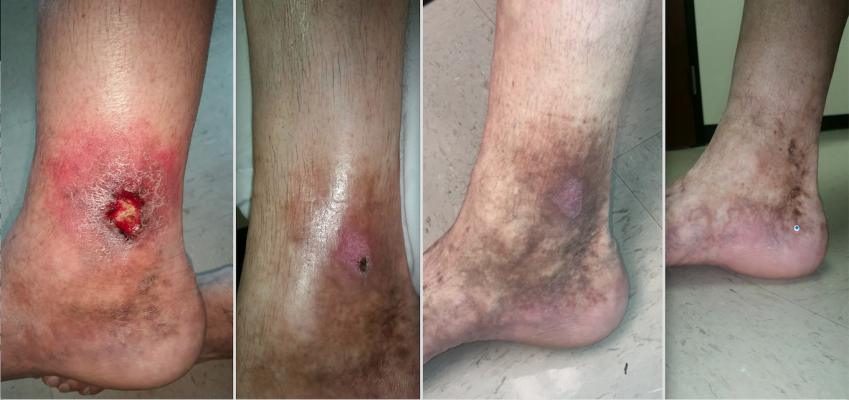
A patient case study showing a venous ulcer formation 18 months following thermal ablation varicose vein treatment in the GSV. The patient was treated with Varithena polidocanol endovenous microfoam chemical-ablation. Images show baseline prior to treatment (right), followed by one, four and six weeks after treatment.
The introduction of thermal ablation revolutionized the treatment of varicose veins, yet recurrence remains a stubborn problem. A 2016 review of three randomized controlled trials (RCTs) involving radiofrequency (RF) ablation and five RCTs employing endovenous laser therapy (EVLT)—all with a minimum of two years’ follow-up data—found 22 percent of patients develop recurrent varicose veins after ablation of the great saphenous vein (GSV). This figure is similar to the recurrence rate in patients undergoing ligature and stripping.
Unfortunately, most patients who develop recurrent varicosities are not candidates for a second round of thermal ablation. Their anatomy simply is not amenable to access from the catheters we use to perform these procedures. Moreover, certain venous conditions that may precede varicose veins can make standard treatment options impractical. However, the FDA’s approval of polidocanol endovenous microfoam (Varithena, BTG) in November 2013, has opened the door to treating the subset of patients whose anatomy makes it difficult and often impossible to treat with traditional options.
Recurrent Venous Reflux Disease
The primary cause of varicose veins is reflux in either the GSV, the anterior saphenous vein, or the small saphenous vein. Because these veins are typically straight for a significant portion of their length, they can accept and be treated with the catheters we use to perform RF and laser ablation.
However, while the catheters used in RF and laser can deliver effective therapy to relatively straight veins, they are somewhat stiff. As a result, one thing they do not do well is make turns. That is significant, because recurrent varicose veins often occur secondary to neovascularization. This phenomenon typically involves Medusa-like formations that start off in the groin and work their way down the thigh before feeding into varicose veins or other segments of veins. Threading a catheter through these very tortuous veins is simply not possible in most cases.
On the other hand, polidocanol endovenous microfoam moves easily through tortuous veins. In the past, I have used 1 or 2 cc’s of physician-compounded foam (PCF), which is polidocanol aerosolized with room air. This is typically for treating very small veins, usually for cosmetic reasons. But for larger veins, Varithena offers a valuable new treatment option. Not only does the preparation effectively seal off dysfunctional veins, but its sterility and safety profile make it a superior choice compared to PCF.
The bubbles that form polidocanol endovenous microfoam are smaller and more uniform than those in PCF, but the difference in the solubility of the two products is critically important, too. The room air used to make PCF contains a lot of nitrogen, about 78 percent. Nitrogen is not very soluble in blood, so when it is injected into circulation, it remains in the form of a gas for a relatively long period. Having nitrogen bubbles circulating throughout the body creates the possibility for air emboli that in a small number of cases have caused adverse events, including strokes, transient ischemic attacks (TIA), and speech and visual disturbances.
By contrast, polidocanol endovenous microfoam contains only a small amount of nitrogen (0.8 percent) and is primarily composed of oxygen and carbon dioxide, which are highly soluble in the blood. Oxygen binds to hemoglobin and carbon dioxide is converted rapidly to bicarbonate before harmlessly entering circulation. We have not had any complications with polidocanol endovenous microfoam in my practice.
Treating GSV With Previous SVT
Another challenging scenario in which polidocanol endovenous microfoam proves valuable is when a patient requires treatment for a varicosity of a GSV that had a previous superficial venous thrombosis (SVT). At least 125,000 Americans experience SVTs each year, though due to underreporting that figure is likely much higher. SVTs usually resolve on their own, as the body clears out the clotting that causes this condition and blood flow resumes. However, an SVT leaves behind scar tissue and other damage in its wake that creates blockages. As a result, even though blood can flow through the GSV after resolution of an SVT, the lumen is not clear enough to pass a guide wire or catheter through and these devices would get stuck.
We see similar changes to the GSV in some patients who have undergone endovascular treatment such as RF or EVLT. Failure of these procedures can result in recanalization of the vein. When that happens, the GSV is typically left with irregular scar tissue within the lumen. As with a prior SVT, a vein that recanalizes following treatment with RF or EVLT is typically not a candidate for a second round of thermal ablation due to these obstructive changes.
However, foam goes wherever blood goes. That means a patient can have these blockages in the GSV, but Varithena flows right around them and can make contact with the lining of the vein.
Varithena Chemical Ablation Case Study
The patient was a male, age 52, who had initially presented with the complaint of heaviness and fatigue in his legs, as well as darkening of the skin just above the inner aspect of both ankles. Both legs also had bulging veins and the patient reported intense itching around his ankles. We treated the patient with bilateral GSV radiofrequency ablations in August of 2015, which significantly improved his symptoms.
However, approximately one year later, the patient noticed a recurrence of similar symptoms in his right leg. Six months later, he also developed an open sore along the medial aspect of the right ankle. When the patient presented for evaluation, we discovered that the GSV had recanalized and had developed a moderate amount of irregular, chronic thrombus. Reflux was noted from the saphenofemoral junction to the level of the ulcer.
At this point, we decided that the best option for this patient was to close his right GSV by performing percutaneous, ultrasound-guided chemical ablation with Varithena. The entire vein was treated in one session with two distinct access points, and the patient was sent home with instructions to wear compression stockings. On follow-up evaluation four days after the procedure, the patient reported that the symptoms of heaviness, pain and itching had resolved. He was instructed to wear compression stockings for at least 14 days to ensure proper healing. An ultrasound performed one week after the procedure demonstrated that the entire length of the GSV was closed and there was no evidence of DVT. At follow-up evaluations four weeks and six weeks later, we noted very expeditious healing of the patient’s venous ulcer. See Figure 1.
A Role in Primary Treatment Using Chemical Ablation
RF ablation and laser ablation remain the treatment of choice for straightforward saphenous veins or small saphenous veins. However, polidocanol endovenous microfoam can have an adjunct role in treating patients with varicosities in these veins. Following thermal ablation, in some patients I will treat the varicose tributaries with either a phlebectomy or polidocanol endovenous microfoam. Moreover, patients with advanced vein disease that has produced ulcers and other skin changes often have a lot of very superficial veins just above the inner aspect of their ankle. Polidocanol endovenous microfoam has been very effective in treating these veins, too. As we all strive to offer the best and most complete treatment options for patients with chronic venous insufficiency, Varithena has been a great addition to my tool box to help ensure safe, accurate and complete treatment.
Editor's note: Tif Siragusa, M.D., is a vascular surgeon at the Siragusa Vein and Laser Center in Nashville, Tenn. He is fellowship trained in the management of vascular disorders with expertise in minimally invasive interventional treatment of vascular disease. During his fellowship training he fine tuned his skills in diagnosing and treating all vascular conditions including varicose veins, carotid artery disease, abdominal aortic aneurysms, and peripheral arterial disease. Siragusa obtained Board Certification in general surgery and vascular surgery. Dr. Siragusa reports no conflicts of interest.
References:

 November 13, 2025
November 13, 2025