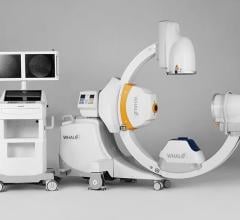
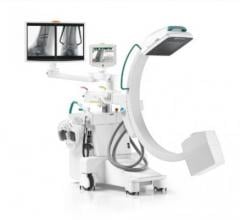
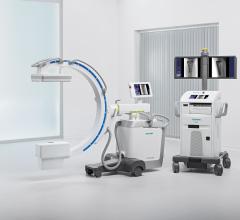
May 5, 2008 — The FDA has notified GE Healthcare’s Surgery Business that the company has satisfied the criteria in the January 2007 consent decree required to resume operations, and can distribute the OEC 9900 Elite C-arm.
The 9900, a fluoroscopy device that uses x-rays to reveal real-time imagery of a patient’s internal structure, is the first product to receive manufacturing and shipping authorization. More than 300 of these units will be shipped to customers in the first 10 days of operations.
After an August 2006 FDA inspection of GE facilities in Salt Lake City and Lawrence, MA, the FDA required that the company suspend C-arm manufacturing and distribution until it met FDA quality management criteria, specific regulations and technical improvements.
Since that time, GE has taken the necessary steps to improve the 9900 and 9800 models, and subsequently filed a new 510(k) submission for the 9900 Elite C-arm “because the company felt it had made enough modifications to the system to warrant a new application,” according to Peter McCabe, president of CEO of GE Healthcare Surgery Business. GE addressed technical issues with the 9900 first because that system is the company's flagship C-arm. The 9800 model is also undergoing the same process, but the company has not said if it will need to file a 510(k) for that system.
McCabe spoke to Imaging Technology News about the improvements it has made to the models and the impact of the suspension – both the bad and the good – on its business.
Imaging Technology News (ITN): What were the steps required by the FDA for GE Healthcare to resume shipment of the 9900 and 9800 Elite C-arms?
Peter McCabe (PM): The consent decree required that we meet three conditions so that we get back to shipment.
We had to create a corrective action plan that spells out how we are going to improve the units that are already out there in the field.
The second thing was we had to have a third party expert review our quality management system and certify that as compliant with the regulations. They also had to review we are making to the product that they are appropriate and effective.
The third thing was the FDA had its opportunity to review our quality management systems and make sure it met the regulations and review the improvements we made to the products to ensure they were appropriate and effective. That is what has been accomplished.
On top that we have gotten approval for the 510K [on the 9900 Elite C-arm], which came through this morning, which is the final straw that lets us get back to shipping.
ITN: What were the types of technical problems that required fixing?
PM: The improvements made to the products were largely on bugs in the system. For example, if you are saving an image, and if you pull the plug out in the middle of saving, you could lose an image or it could get mixed up. What we’ve done is put in an uninterruptible power supply or UPS so that if somebody pulls the plug, there’s still power, and that image is not lost. So we fixed issues like that.
We would like to think that our products were pretty good yesterday, but those products will more than double the reliability and make it far and away the most reliable product we’ve ever put out in the history of our business.
ITN: Were these enhancements made to all of the Elite models?
PM: Only the 9900 right now. We are only able to ship now the 9900. We’re going through a similar process for the 9800, and that will be time delayed.
Our 9900 is our premier product that fundamentally replaces the 9800. But we also want to make these improvements in the 9800 and get them out there.
ITN: Were there recalls made on the 9900 Elite and 9800 models?
PM: There were recalls. What it ends up being is a notification to the customer that says, we are working on a technical fix, and when the technical fix comes through, we will implement it for free on your unit.
ITN: How long will it be before the 9800 updates can be implemented?
PM: We are in the final stages of developing and testing. I would anticipate it is going to be toward that latter part of this year.
ITN: The suspension cost GE hundreds of millions of dollars in lost revenues and in expenses. What is the estimated cost to GE as a result of the suspension?
PM: Fundamentally, you’re operating a business for 20 months that is bringing in no product revenue. You’re paying fee and all of the quality engineers and all of the quality guys who are working on it. So you have this big sink of cost. Then you have a big loss of revenue in parallel with that. But we were still running our service business, so that was not zero revenue. Throw those two together and you frame a loss of hundreds of millions of dollars. At this point it is water under the bridge for future of the business.
ITN: Why do you think the hospitals were willing to wait for the delivery of the C-Arms?
PM: Our customers are loyal to us because they say ‘you have product quality.’ Then you throw in your service and your clinical applications support. So, customers have grown to really love the products, and fundamentally it is the image quality, the simplicity and the overwhelming support we give with field and clinical folks.
The 9900 comes in the OEC Elite 9900 Ortho/Surgery and the OEC Elite 9900 Vascular configurations.
The 9800 comes in the OEC 9800 Plus Ortho/Surgery and the OEC 9800 Plus Vascular configurations.


 June 20, 2024
June 20, 2024