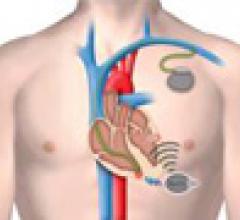
March 6, 2012 — Medtronic Inc. announced the launch of the CapSure Sense MRI SureScan pacing leads, which are approved ...
Leads Implantable Devices
News and new technology innovations for leads implantable devices can be found on this channel.
January 31, 2012 — Medtronic Inc. announced U.S. Food and Drug Administration (FDA) approval and launch of the DF4 High ...
January 26, 2012 — St. Jude Medical recently announced U.S. Food and Drug Administration (FDA) approval of its Unify ...
December 16, 2011 – St. Jude Medical began a voluntary recall Nov. 28 for its Riata and Riata ST Silicone Defibrillation ...
November 16, 2011 – Cambridge Consultants unveiled how it has collaborated with start-up company EBR Systems to develop ...
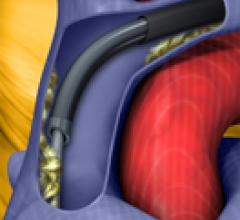
May 4, 2011 – A new version of the Liberator Locking Stylet with Beacon Tip Technology, a distinct radiopaque tip marker ...
October 15, 2010 – Medtronic has entered into a $268 million settlement relating to its Sprint Fidelis family of ...

Cardiac rhythm device reliability and the approach to managing complications associated with these devices have recently ...
June 1, 2010 – A quadripolar pacing system was recently released in Europe and offers physicians the ability to ...
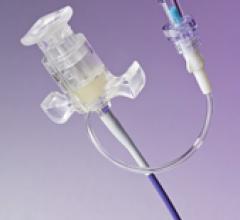
May 13, 2010 – An integrated, break-away hemostasis valve is integrated into a new lead delivery system is ...
May 13, 2010 – A new simulation system to augment traditional procedural training for physicians on laser ...
March 22, 2010 – A pivotal clinical trial recently began to gain U.S. approval for a defibrillator system that ...
February 3, 2010 – Results of a four-year study evaluating the safety and efficacy of laser assisted lead removal ...
December 8, 2009 – The U.S. Food and Drug Administration (FDA) gave 510(k) clearance last week for Spectranetics’ ...
November 12, 2009 – The first enrollments were made this week in the global Attain Success trial that will ...

 March 06, 2012
March 06, 2012