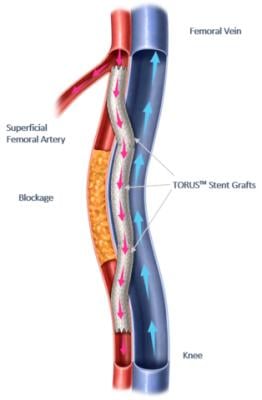
Percutaneous Transmural Arterial Bypass (PTAB) using the DETOUR System (Graphic: Business Wire)
June 15, 2023 — Endologix LLC, a privately held, global medical device company dedicated to providing disruptive therapies for the interventional treatment of vascular disease, announced that the U.S. Food and Drug Administration (FDA) has granted approval for the DETOUR System to treat patients with complex peripheral arterial disease (PAD).
Over 8.5 million Americans are affected by PAD. This condition can cause significant pain, reduced mobility, and even amputation. At present, patients with long blockages of the superficial femoral artery (SFA) have limited treatment options. The recommended therapy is open surgical bypass, which is invasive, has a high early complication rate, and requires prolonged recovery. Endovascular treatments are feasible in selected patients but have limited patency.
Percutaneous Transmural Arterial Bypass (PTAB) with the DETOUR System offers a novel approach to treating complex PAD, enabling physicians to bypass lesions in the superficial femoral artery by using stents routed through the femoral vein to restore blood flow to the leg. This approach is particularly effective for patients with long lesions (20cm-46cm in length), those that have already undergone failed endovascular procedures, or those that may be sub-optimal candidates for open surgical bypass.
"We are delighted to receive FDA approval of the DETOUR System," said Matt Thompson, MD, President, and CEO of Endologix. “PTAB therapy represents a significant step forward for patients with complex PAD, they have long needed a more effective and less invasive treatment option for long lesions of the SFA. We are proud to be pioneering this novel approach and continuing to innovate on behalf of patients. We look forward to launching this new therapy in the U.S. through a targeted market release in the coming weeks."
With the introduction of the PTAB therapy, Endologix continues to build on its legacy of disruptive vascular therapies to improve patient outcomes. The DETOUR System represents a breakthrough in the treatment of PAD, and the company is committed to focusing on patients with the most pressing unmet clinical needs.
For more information: www.endologix.co


 November 24, 2025
November 24, 2025 









