May 26, 2026 — According to researchers in Europe, women are more likely to face delays in diagnosis of cardiovascular disease and, as a result, they are more likely to die or develop more serious ...
Peripheral Artery Disease (PAD)
This channel includes news, interventions, and new technology innovations for peripheral artery diease, PAD and critical limb ischemia.
May 26, 2026 — According to researchers in Europe, women are more likely to face delays in diagnosis of cardiovascular ...
March 30, 2026 — Helen H. Hobbs, M.D., professor in the Eugene McDermott Center for Human Growth and Development and of ...
March 18, 2026 — Circle Cardiovascular Imaging (Circle CVI) has announced the expansion of its cvi42 cardiovascular ...
March 17, 2026 — Empassion Health, Inc. has announced a strategic partnership with Karoo Health Inc, America’s Cardiac ...
March 4, 2026 — Inflo Health has launched a comprehensive cardiology suite designed to address care management gaps ...
Feb. 14, 2026 — Cardiovascular Research Foundation (CRF) has announced the creation of Complex Coronary Summit, a new ...
Jan. 27, 2026 — Robocath has launched the world’s first FIH (First-In-Human) clinical study evaluating its new robotic ...
Dec. 10, 2025 — Medtronic plc has announced the first commercial use of the Liberant thrombectomy system (Liberant) ...
Dec. 15, 2025 — Royal Philips has entered into an agreement to acquire SpectraWAVE, Inc., an innovator in enhanced ...
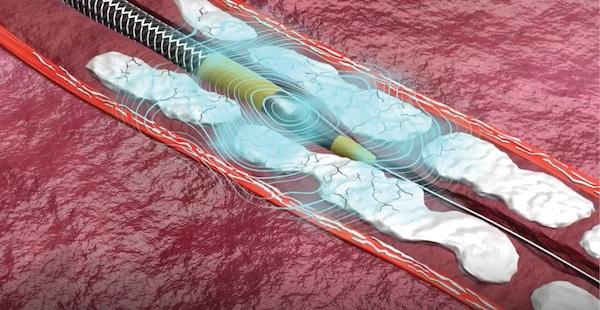
Since receiving FDA approval in 2016, intravascular lithotripsy (IVL) systems have grown in popularity among ...
Nov. 4, 2025 — Amplitude Vascular Systems (AVS), a medical device company focused on treating calcified arterial disease ...
Nov. 4, 2025 – Johnson & Johnson MedTech has announced the one-year results in patients treated with its Shockwave ...
Sept. 9, 2025 — Amplitude Vascular Systems (AVS), a medical device company focused on safely and effectively treating ...
June 6, 2025 – A large retrospective review of more than 100,000 patients found the use of glucagon-like peptide-1 ...
Feb. 3, 2025 — Today, Peripheral Artery Disease (PAD) affects 10 million Americans and is the most common cause of limb ...


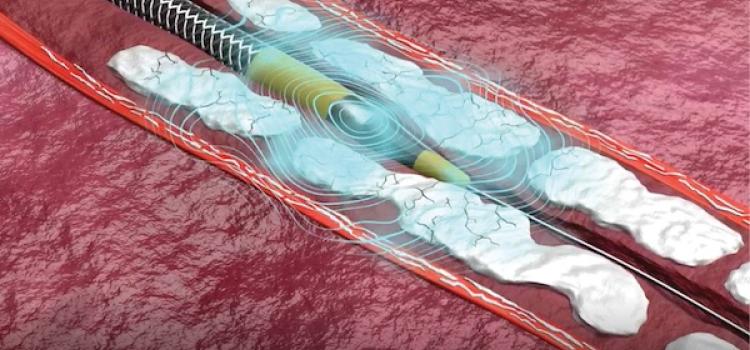



 May 26, 2026
May 26, 2026