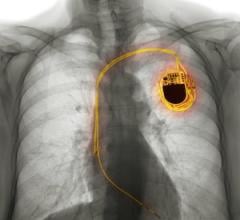
April 24, 2026 — A new analysis from the NOAH – AFNET 6 trial shows that artificial intelligence (AI) can help unlock data from pacemakers to better understand stroke risk in patients with device ...
Atrial Fibrillation
This channel includes news and new technology innovations for the treatment of atrial fibrillation, also referred to as AF or afib. AF is a cardiac arrhythmia caused by irregular and often rapid heart rate. It is caused by the upper chambers (the atria) beating irregularly and uncoordinated with the lower ventricle chambers of of the heart. Symptoms include weakness with heart palpitations and shortness of breath. The conditional can lead to an increased risk of stroke and heart failure. AF episodes can cause the blood in the atria to stagnate and form clots, usually within the left atrial appendage (LAA). The clots can flow to the brain and cause a stroke. Treatments include anticoagulation therapy to dissolve clots, catheter or surgical ablation and LAA occlusion.
April 24, 2026 — Johnson & Johnson recently announced the initiation of the PERSIGMA randomized controlled trial (RCT) ...
April 24, 2026 — A new analysis from the NOAH – AFNET 6 trial shows that artificial intelligence (AI) can help unlock ...
April 24, 2026 — The Heart Rhythm Society has announced the first analysis of its Centers of Excellence Optimal ...
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...
April 23, 2026 — A new study analyzing data from more than 13,000 patients found that people using the increasingly ...
April 13, 2026 — Heart failure is common in people who have atrial fibrillation detected during screening, according to ...
April 13, 2026 — Johnson & Johnson has announced favorable 12-month interim effectiveness and safety results from ...
For over a decade, the cardiac cryoablation industry has seen little in the way of technological advancements. Yet ...
April 2, 2026 — Omron Healthcare Co. has launched a randomized controlled trial in collaboration with the University of ...
Feb. 9, 2026 — Argá Medtech has announced positive clinical results from the BURST-AF (NCT05572047) first-in-human trial ...
Feb. 9, 2026 — Atraverse Medical presented new clinical and preclinical data at AF Symposium 2026 in Boston, further ...
In the United States, the options currently available for cardiac ablation use thermal mechanisms to ablate tissue and ...
Feb. 6, 2026 — Abbott has announced new clinical data from two late-breaking presentations at AF Symposium in Boston ...
Feb. 3, 2026 — Bristol Myers Squibb has launched "Change the Target. Change What’s Possible," an educational campaign ...
Jan. 20, 2026 — Abbott has received CE Mark in Europe for the TactiFlex Duo Ablation Catheter, Sensor Enabled to treat ...
When the patients of Michael Boler, M.D. need cardiac monitoring, the Holter monitor is no longer his first choice. “The ...
Jan. 20, 2026 — Kardium Inc. has announced the publication of the PULSAR clinical trial results in the Journal of the ...
Dec. 19, 2025 — Johnson & Johnson MedTech has announced its sponsorship of a new data collection platform developed by ...
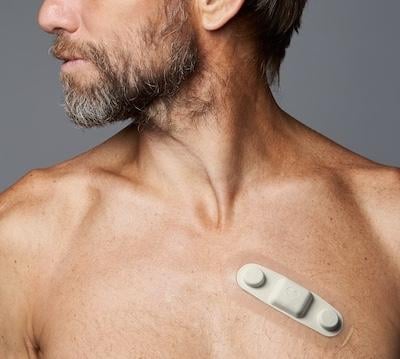
Atrial fibrillation (AFib) is one of the most common heart rhythm disorders, but researchers still don’t have a reliable ...

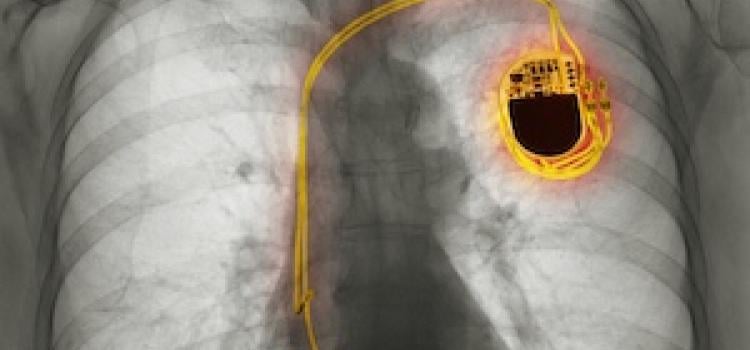



 April 27, 2026
April 27, 2026