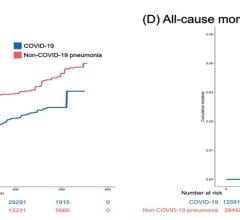
April 24, 2026 – Findings from the North American COVID-19 Myocardial Infarction (NACMI) registry demonstrate significantly higher one-year mortality rates in patients with COVID-19 and ST-elevation ...
Coronavirus (COVID-19)
This page contains medical information for clinicians on the 2019 Novel Coronavirus (COVID-19, also called 2019-nCoV, and now clinically SARS‐CoV‐2). This section includes articles that pertain to clinicians and cardiologists on the virus, new technologies being deployed to fight the virus and clinical information from various sources. Here are direct links for medical professionals to COVID-19 resources from the U.S. Food and Drug Administration (FDA), Centers for Disease Control (CDC) and the World Health Organization (WHO). Daily world-wide statistics on the coronavirus outbreak are available from the WHO Situations Reports. Here is the Centers for Medicare and Medicaid Services (CMS) frequently asked questions and answers (FAQs) for healthcare providers regarding Medicare payment for laboratory tests and other services related to the COVID-19.
April 24, 2026 – Findings from the North American COVID-19 Myocardial Infarction (NACMI) registry demonstrate ...
Feb. 11, 2026 —The American Roentgen Ray Society (ARRS) has announced the following radiologists, as well as their ...
July 25, 2025 — Data in recent staffing surveys from the American Society of Radiologic Technologists show that vacancy ...
During the COVID-19 pandemic, many cardiology departments were faced with the daunting task of supporting inpatient and ...
July 21, 2025 — Long COVID patients with abnormal cardiopulmonary PET/MR findings may be more likely to develop heart ...
March 20, 2024 — SARS-CoV-2, the virus that causes COVID-19, can damage the heart even without directly infecting the ...

It is a new year with lots of new content! Here is a look at the most-read content during the month of January on dicard ...
COVID-19 has posed challenges for physicians whose cardiac patients are at-risk and reluctant to schedule an office ...
January 25, 2024 — New research confirms what public health leaders have been fearing: the significant uptick in the ...
January 11, 2024 — Acute cardiovascular manifestations of COVID-19, such as heart failure, thrombosis, and dysrhythmia ...

Here is a rundown of what you and your colleagues found to be most interesting in the field of diagnostic and ...
December 14, 2023 — Individuals infected with COVID-19 are also at an increased risk of suffering from heart rhythm ...
November 20, 2023 — In the U.S., nearly 10,000 children and adolescents have developed a condition called multisystem ...

As another month comes to a close, DAIC takes a look at the Top 10 most-read pieces of content from www.dicardiology.com ...
October 16, 2023 — A door-to-balloon (D2B) time of 90-minutes or less is associated with improved outcomes for heart ...
October 11, 2023 — Unvaccinated people who have recovered from COVID-19 are at heightened risk of heart complications ...
September 26, 2023 — New research from the Smidt Heart Institute at Cedars-Sinai shows that patients who went to a ...






 April 27, 2026
April 27, 2026