April 6, 2022 – The National Institute of Health awarded $436,642 to associate professor of biomedical engineering ...
Coronavirus (COVID-19)
This page contains medical information for clinicians on the 2019 Novel Coronavirus (COVID-19, also called 2019-nCoV, and now clinically SARS‐CoV‐2). This section includes articles that pertain to clinicians and cardiologists on the virus, new technologies being deployed to fight the virus and clinical information from various sources. Here are direct links for medical professionals to COVID-19 resources from the U.S. Food and Drug Administration (FDA), Centers for Disease Control (CDC) and the World Health Organization (WHO). Daily world-wide statistics on the coronavirus outbreak are available from the WHO Situations Reports. Here is the Centers for Medicare and Medicaid Services (CMS) frequently asked questions and answers (FAQs) for healthcare providers regarding Medicare payment for laboratory tests and other services related to the COVID-19.
April 4, 2022 – Keck Medicine of USC has launched its COVID Recovery Clinic, a multidisciplinary program led by a core ...
April 1, 2022 – Over the course of the pandemic, a number of studies examined whether COVID-19 patients were at ...
During the COVID-19 pandemic, many cardiology departments were faced with the daunting task of supporting inpatient and ...
March 31, 2022 – A study conducted at São Paulo State University (UNESP) in Brazil shows that even mild to moderate ...
March 29, 2022 — Today the U.S. Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention ...
March 24, 2022 — Royal Philips announced its latest introductions in analytics and interoperability solutions at the HIM ...
COVID-19 has posed challenges for physicians whose cardiac patients are at-risk and reluctant to schedule an office ...
March 22, 2022 — A small study has shown that SARS-CoV-2 vaccination in patients who had an inflamed heart muscle in the ...
March 13, 2022 – People with a congenital heart defect who were hospitalized with COVID-19 infection were at higher risk ...
March 11, 2022 – A new study by physicians at Columbia University Vagelos College of Physicians and Surgeons has ...
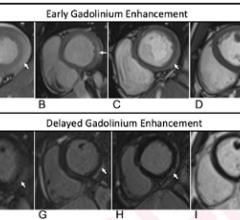
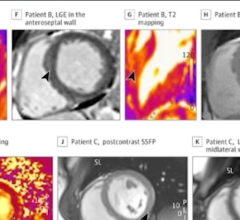
February 17, 2022 — While cardiac imaging shows that COVID-19 vaccine–associated myocarditis has a similar pattern as ...
February 15, 2022 — Vaccine-associated myocarditis shows a similar injury pattern on cardiac MRI compared to other ...

The latest cardiology practice-changing scientific breakthrough, late-breaking study presentations have been announced ...
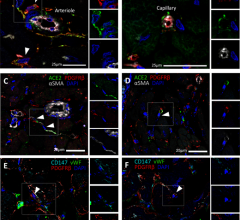
February 9, 2022 — A new study has shown how SARS-CoV-2 may contribute to severe microvascular damage seen in severely ...
Campbell Rogers, M.D., chief medical officer of HeartFlow, explains how hospitals are using CT image-based fractional ...
Siddharth Singh, M.D., director of the COVID-19 heart program, staff cardiologist and echocardiographer, Cedars-Sinai ...


 April 06, 2022
April 06, 2022