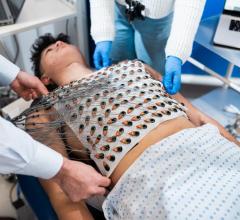
May 14, 2025 — BrightHeart, a provider of artificial intelligence (AI) solutions for pediatric cardiology and obstetrics, recently announced it has received 510(k) clearance from the U.S. Food and ...
Congenital Heart
This Congenital Heart Disease channel includes news and new technology to diagnose, treat and manage congenital heart disease. This includes transposed vessels, tetralogy of Fallot, pediatric cardiomyopathy, pulmonary heart valves, pulmonary atresia, patent foramen ovale (PFO), atrial septal defect (ASD) and ventricular septal defect (VSD).
May 14, 2025 — BrightHeart, a provider of artificial intelligence (AI) solutions for pediatric cardiology and obstetrics ...
March 31, 2025 — AGEPHA Pharma USA, LLC, has highlighted clinical data that provides new information about how low-dose ...
March 17, 2025 — Being born with a heart defect may be associated with an increased cancer risk for babies and their ...
June 20, 2024 — atHeart Medical, a medical device company establishing a new standard of care for atrial septal defects ...
December 19, 2023 — A vest that can map the electrical activity of the heart in fine detail could potentially be used to ...
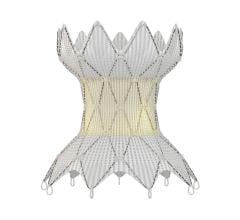
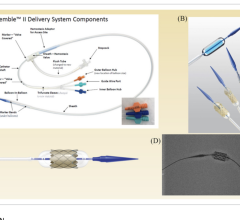
October 25, 2023 — For its novel multi-lumen stent system for interventional adjustment of pulmonary blood flow in ...
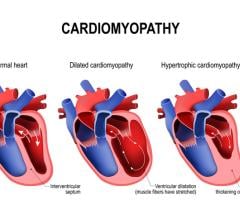
August 31, 2023 — The European Society of Cardiology (ESC) Guidelines on cardiomyopathies are published online in Europe ...
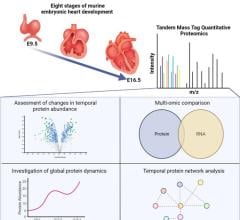
July 3, 2023 — Researchers at the UNC School of Medicine, UNC McAllister Heart Institute, and the UNC Lineberger ...
February 6, 2023 — Recent advances in newborn heart surgery have greatly reduced brain injuries in infants with ...
January 27, 2023 — Cincinnati Children’s Hospital Medical Center is excited to announce the opening of its Heart and ...
January 24, 2023 — Cardiothoracic surgeon Thomas E. MacGillivray, MD, from MedStar Health, was elected President of The ...
January 6, 2023 — Patients with congenital heart diseases often suffer from obstructions in the right ventricular ...




 May 14, 2025
May 14, 2025