April 24, 2026 – A novel detection algorithm spotted moderate-to-severe aortic stenosis (AS) with a sensitivity of 90.5% of all patients and 100% of African American patients. Researchers presented ...
Heart Valve Technology
This channel includes news and new device innovations about heart valve technologies, including the aortic valve, mitral valve, pulmonic valve, and tricuspid valve. This includes information on transcatheter valve technologies like transcatheter aortic valve replacement (TAVR, or implantation TAVI), transcatheter mitral valve repair or replacement (TMVR), transcatheter and surgical valve repairs, and surgical replacement valves. Newer devices are now being used for transcatheter tricuspid valve repair replacement (TTVR).
April 24, 2026 – A novel detection algorithm spotted moderate-to-severe aortic stenosis (AS) with a sensitivity of 90.5% ...
April 21, 2026 — A new multicenter study published in The Annals of Thoracic Surgery finds that reoperative surgical ...
April 7, 2026 —TRiCares SAS has received approval from the U.S. Food and Drug Administration (FDA) for an ...
March 31, 2026 — Solo Pace Inc. has announced the commercial release of the SoloPace Fusion Temporary Pacing System. The ...
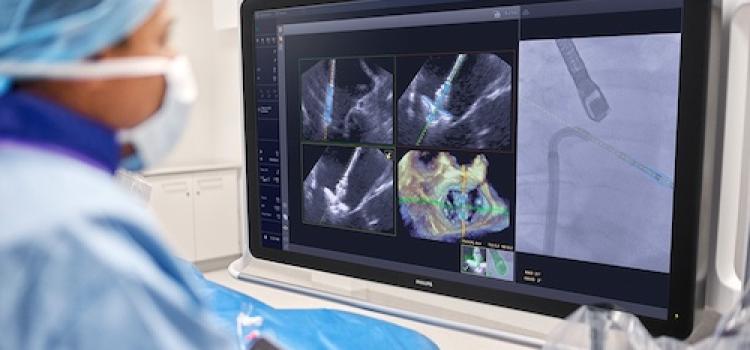
March 25, 2026 — Royal Philips has received U.S. Food and Drug Administration (FDA) 510(k) clearance for EchoNavigator ...
Feb. 5, 2026 — New research shows that the use of an AI-enabled digital stethoscope more than doubled the identification ...
Dec. 26, 2025 — Edwards Lifesciences has announced the company’s SAPIEN M3 mitral valve replacement system, a ...
Nov. 17, 2025 — Royal Philips has introduced DeviceGuide, an AI-powered device tracking* solution that assists ...
Oct. 28, 2025 — People who underwent a minimally invasive procedure to have their heart’s aortic valve replaced had ...
Oct. 25, 2025 — Medtronic plc has announced the launch of the Stedi Extra Support guidewire, designed to enhance ...
Aug. 28, 2025 — Medtronic plc has announced it received U.S. Food and Drug Administration (FDA) approval for the ...
Aug. 18, 2025 — A new study published in The Annals of Thoracic Surgery, a journal from The Society of Thoracic Surgeons ...
Aug. 4, 2025 — Corcym announced that its Perceval Plus sutureless aortic heart valve was used in a first-ever robotic ...
July 10, 2025 — On July 2, 2025, Centers for Medicare & Medicaid Services (CMS) issued a National Coverage Determination ...
June 27, 2025 – Foldax Inc., a leader in the development of innovative polymer heart valves, has announced compelling ...




 April 24, 2026
April 24, 2026