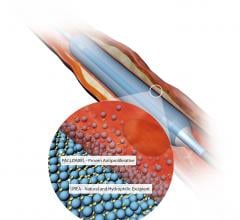
November 3, 2020 — The U.S. Food and Drug Administration (FDA) has cleared the Boston Scientific Ranger Drug-coated ...
Peripheral Artery Disease (PAD)
This channel includes news, interventions, and new technology innovations for peripheral artery diease, PAD and critical limb ischemia.
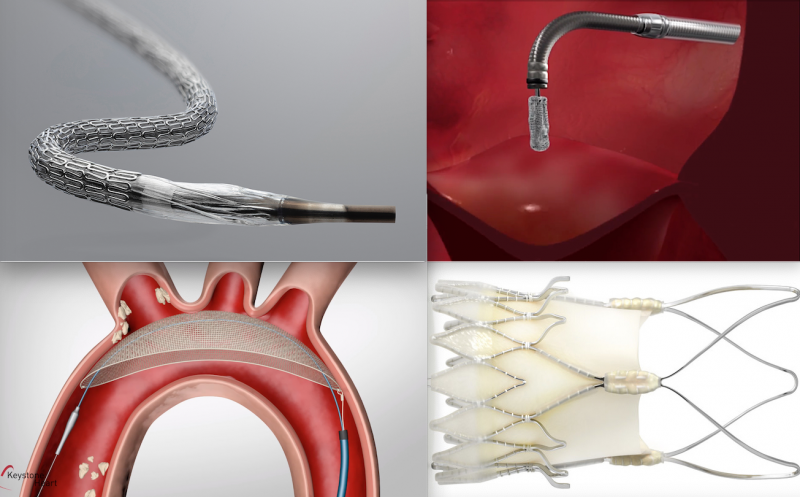
Here are some of the key takeaways from the late-breaking interventional cardiology and structural heart trials ...

October 18, 2020 – A large subgroup analysis of a randomized clinical trial showed neither a mortality risk nor benefit ...
October 18, 2020 – The first results from the IN.PACT Below the Knee (BTK) Study, a feasibility study assessing the ...
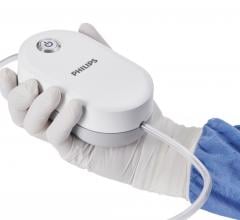
September 25, 2020 — Philips Healthcare launched its QuickClear mechanical thrombectomy system. The compact, single-use ...
September 14, 2020 — Avinger received U.S. Food and Drug Administration (FDA) 510(k) clearance from the FDA for its ...
September 4, 2020 — The U.S. Centers for Medicare and Medicaid Services (CMS) granted a New Technology Add-on Payment ...
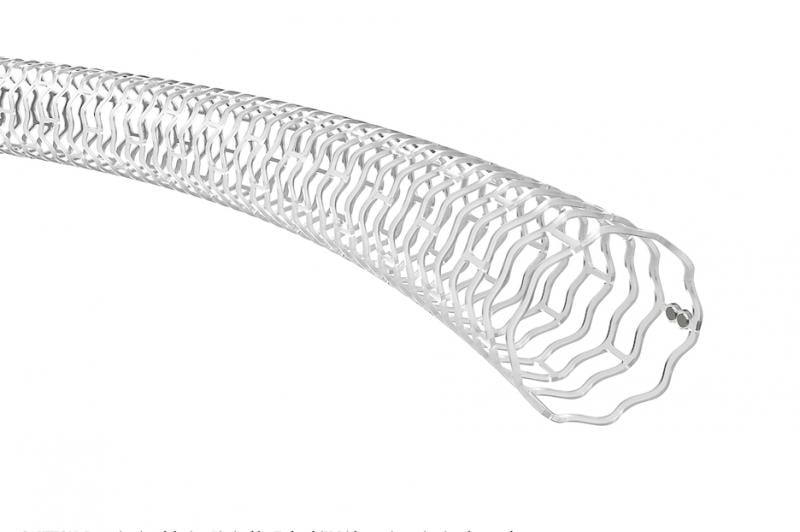
September 3, 2020 — Abbott today announced the start of the LIFE-BTK clinical trial to evaluate the safety and ...
July 20, 2020 – BD (Becton, Dickinson and Company) recently completed the acquisition of Straub Medical AG, a privately ...
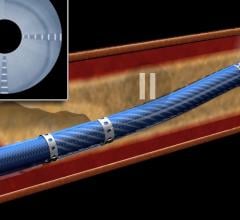
June 16, 2020 — The 36-month results from Veryan Medical’s MIMICS-2 study for the BioMimics 3D femoropopliteal stent ...
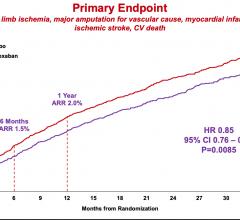
March 28, 2020 — People with symptomatic peripheral artery disease (PAD) who took the blood thinner rivaroxaban with ...
March 20, 2020 — The Centers for Medicare and Medicaid Services (CMS) announced March 18, 2020, that all elective ...
February 13, 2020 — The U.S. Food and Drug Administration (FDA) has issued its final guidance on "Peripheral Vascular ...

Cardiovascular diseases (CVDs) are among the leading causes of death across the globe. For patients suffering from high ...

December 16, 2019 — Here is the list of the most popular content on the Diagnostic and Interventional Cardiology (DAIC) ...


 November 03, 2020
November 03, 2020