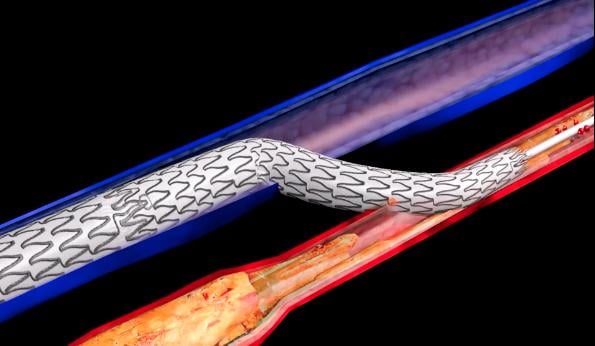
The LimFlow procedure permanently bypasses unreconstructable arteries and leverages healthier veins as a conduit to create new routes to perfuse tissue in the foot.
November 14, 2019 — Results were positive for the multicenter PROMISE I Trial represents the first human use in the United States of a purpose-built percutaneous deep vein arterialization system for the treatment of no-option chronic limb-threatening ischemia (CLTI) patients. Data was presented at the 2019 Vascular Interventional Advances (VIVA) annual meeting.
The LimFlow procedure permanently bypasses unreconstructable arteries and leverages healthier veins as a conduit to create new routes to perfuse tissue in the foot. The purpose of the PROMISE I trial is to establish the safety, effectiveness, and feasibility of the LimFlow system for use in the treatment of CLTI. Use of the LimFlow procedure had a technical success rate was 97%, with only one technical failure. The primary safety endpoint of above-ankle amputation-free survival (AFS) at 30 days was 91%. The secondary endpoint of AFS at six months was achieved in 74%, said Daniel Clair, M.D., chair of the Department of Surgery at Palmetto Health-USC Medical Group and senior medical director for Surgical Services at Prisma Health–Midlands.
Thirty-two no-option CLTI patients (mean age, 71 ± 14 years; 66% male) were enrolled in a nonrandomized manner at seven centers across the United States. All enrolled patients had Rutherford class 5 or 6 disease and were deemed by an independent review committee of experts to be ineligible for endovascular or surgical procedures to restore blood flow. Patients underwent percutaneous deep vein arterialization using the LimFlow system.
Find information on all the VIVA 2019 Late-breaking Clinical Trials


 May 06, 2026
May 06, 2026 









