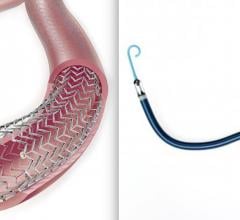
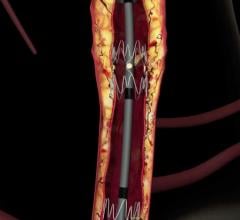
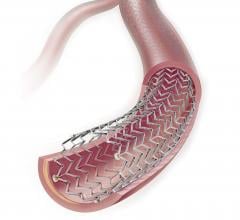
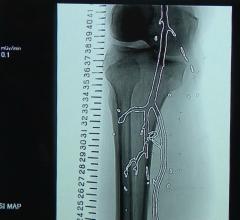
June 20, 2019 – VEITHsymposium and the Cardiovascular Research Foundation (CRF) announced an alliance between Transcathe ...
Peripheral Artery Disease (PAD)
This channel includes news, interventions, and new technology innovations for peripheral artery diease, PAD and critical limb ischemia.
May 29, 2019 — Philips announced the three-year results from the ILLUMENATE Pivotal trial and the ILLUMENATE European ...
April 15, 2019 – Intact Vascular Inc. received U.S. Food and Drug Administration (FDA) market clearance for the Tack ...
February 27, 2019 — Ra Medical Systems Inc. announced a 98 percent success rate in the results from a 52-patient study u ...

The Society for Cardiovascular Angiography and Interventions (SCAI) 2019 Scientific Sessions in Las Vegas, May 19-22 ...
Clinical study data makes the world go around in cardiology and is the basis of setting guidelines in evidence-based ...
February 12, 2019 — A study presented at the 2018 annual meeting of the Cardiovascular and Interventional Radiology ...

In recent years, there has been a lot of focus by vendors on developing better stenting technologies to treat peripheral ...
January 28, 2019 — Philips announced the latest pooled analysis of patient-level data of over 2,300 patients treated ...
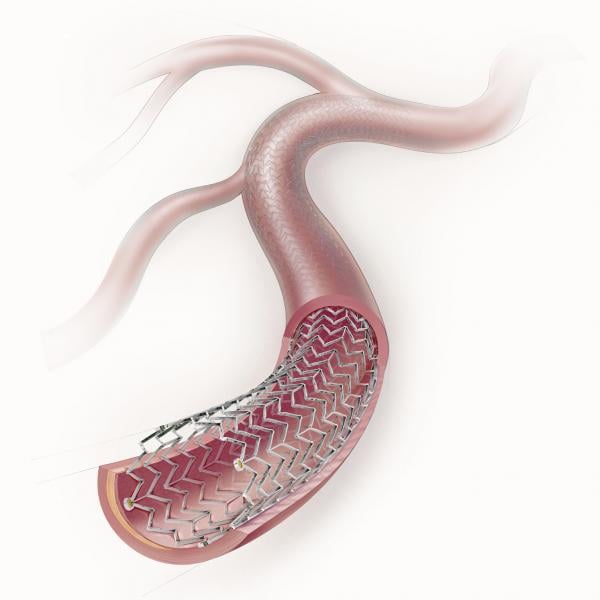
The anti-proliferative drug paclitaxel has been used as a coating on coronary stents to prevent restenosis since 2003 ...
January 25, 2019 — Profusa announced promising clinical data from two studies evaluating the company's Lumee Oxygen ...
January 23, 2019 — A new report in the Journal of Vascular Surgery chronicles a multi-site randomized controlled trial c ...
January 17, 2019 — The U.S. Food and Drug Administration (FDA) issued a letter Jan. 17, 2019, to healthcare providers ...
December 19, 2018 — A new set of appropriate use criteria (AUC) released Dec. 17 by a group of cardiovascular ...
December 18, 2018 — Merit Medical Systems Inc. announced that it has acquired substantially all of the assets of ...

 June 20, 2019
June 20, 2019