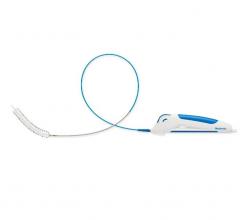
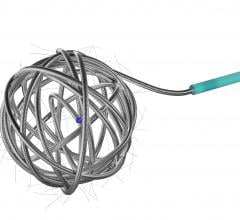
February 1, 2018 – LimFlow SA, developer of minimally-invasive technology for the treatment of end-stage critical limb ...
Peripheral Artery Disease (PAD)
This channel includes news, interventions, and new technology innovations for peripheral artery diease, PAD and critical limb ischemia.
January 26, 2018 – Medtronic plc announced the initiation of its investigational device exemption (IDE) study for the ...
January 24, 2018 — Cardiovascular Systems Inc. recently announced two new partnerships broadening the company’s product ...
January 9, 2018 – Avinger Inc. announced that Arne Schwindt, M.D., a vascular surgeon at St. Franziskus Hospital in ...
December 29, 2017 — iVascular announced the release of the new Oceanus 14 Pro percutaneous transluminal angioplasty (PTA ...

As debates about the current state and future of healthcare rage in Congress, the media and healthcare settings across ...
November 20, 2017 — Avinger Inc. announced in October that the company received 510(k) clearance from the U.S. Food and ...
October 16, 2017 — Medtronic announced the launch of the Concerto 3-D Detachable Coil System at the Cardiovascular and ...
September 29, 2017 — Avinger Inc. recently announced Conformité Européenne (CE) Marking approval for treating in-stent ...
September 22, 2017 — Physicians demonstrated that reducing metal burden in superficial femoral artery (SFA) therapy ...
September 20, 2017 — Not-for-profit preclinical research institute CBSET announced that its scientists have published ...
September 20, 2017 — Shockwave Medical reported positive results from the DISRUPT BTK Study, which were presented at the ...
September 18, 2017 — Philips announced the two-year results from the ILLUMENATE European randomized clinical trial (EU ...

September 14, 2017 — Here are quick summaries for all the key late-breaking vascular and endovascular clinical trials ...
September 13, 2017 — Philips announced its presence at the Vascular Interventional Advances (VIVA 17) Annual Conference ...


 February 01, 2018
February 01, 2018