August 9, 2017 — Ra Medical Systems announced that the United States Patent and Trademark Office (USPTO) has granted the company a broad patent (No. 9700655) that covers its DABRA catheter for cardiovascular disease treatment. The patent was granted on July 11, 2017, and assigned to Ra Medical Systems. The inventors include Ra Medical Systems Director of Research and Development, James Laudenslager, Ph.D., and CEO/CTO Dean Irwin. DABRA (Destruction of Arteriosclerotic Blockages by laser Radiation Ablation) recently received U.S. Food and Drug Administration (FDA) market clearance and is now available in the U.S. and Europe.
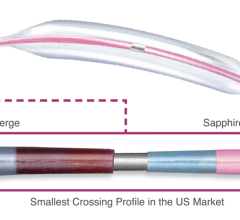
The patent, which covers DABRA’s “small flexible liquid core catheter for laser ablation in body lumens and methods for use,” is pivotal for Ra Medical Systems’ intellectual property, as it provides wide-ranging patent protection for the DABRA catheter. The DABRA catheter uses a liquid fill with a solid window that shows improved tissue removal, compared to other multi-fiber excimer catheters that have a high percentage of dead space. This improved performance contributes to DABRA’s high success rate and low adverse event rate, according to the company.
DABRA treats peripheral artery disease (PAD), the leading cause of limb amputations. The device has shown shorter procedure times, is less expensive and has an impressive safety profile, according to Ra Medical Systems. DABRA may also reduce the costs that are associated with treating PAD and ultimately lead to greater patient access to, and success with, interventional procedures instead of limb amputation.
For more information: www.ramed.com


 November 08, 2024
November 08, 2024