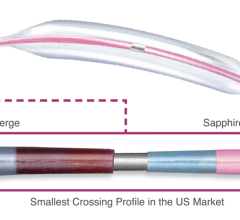
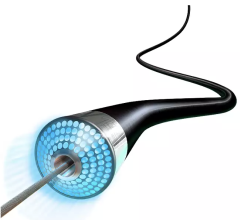
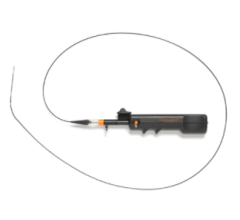
The AngioDynamics/Eximo Medical B-Laser atherectomy system.
November 7, 2019 — There was no difference between drug-coated balloons (DCB) vs. plain old balloon angioplasty (POBA) following B-Laser atherectomy, according to late-breaking study results from the DCB Versus POBA After B-Laser Atherectomy Trial at the Vascular InterVentional Advances (VIVA) Annual Meeting. The results were presented by John H. Rundback, M.D., director of the Interventional Institute at Holy Name Medical Center, Teaneck, N.J.
The AngioDynamics/Eximo Medical B-Laser represents a new atherectomy device utilizing a 355 nm laser with a very short pulse width to selectively ablate mixed-morphology plaque including calcification. Outcomes were compared for patients treated in the European Union CE mark and United States investigational device exemption studies with B-Laser, followed by either POBA (n = 46) or DCB angioplasty (n = 51).
Clinical characteristics were similar between the two groups. POBA patients had more popliteal and tibial lesions treated (77.8 and 84.2% vs. 11.1 and 15.8% in the DCB group), but other lesion characteristics were similar. There were no major procedural complications, including no embolization and no grade C–E dissections. Core lab–adjudicated duplex patency at six months was 89.5% in the POBA group and 85.2% in the DCB group. One-year clinical improvement (ankle-brachial index, Rutherford score) were the same.
These preliminary data indicate no differences in measurable short-term outcomes following B-Laser atherectomy regardless of subsequent mode of angioplasty. The characteristics of plaque modification following B-Laser atherectomy may impact outcomes and mitigate previously noted patency and clinical benefits of DCB angioplasty.
Find information on all the VIVA 2019 Late-breaking Clinical Trials


 November 08, 2024
November 08, 2024