September 13, 2019 — Rex Medical L.P. has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the Revolution Peripheral Atherectomy System.
Regulatory clearance was based on data provided by the REVEAL IDE clinical trial. The results of this pivotal trial demonstrated the safety and effectiveness of the Revolution Peripheral Atherectomy System for lower extremity peripheral arterial revascularization in 121 subjects with femoropopliteal/tibial occlusive disease. The trial met all prespecified primary and secondary safety and effectiveness endpoints.
The Revolution device is indicated for atherectomy of the peripheral vasculature, and to break apart and remove thrombus from the peripheral arteries in patients with occlusive atherosclerotic disease.
Peripheral artery disease (PAD) and its most severe form, critical limb ischemia (CLI), affects more than 10 million people in the U.S. It occurs when an artery becomes blocked or narrowed due to plaque or calcium deposits, which in turn reduces blood flow to the lower extremities. Left untreated, this can result in pain, ulceration, gangrene and amputation. Successful revascularization and blood flow restoration is essential in the treatment and quality of life improvement for this patient population.
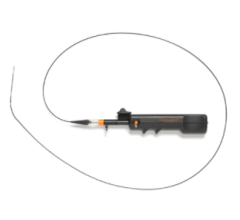
The Rex Medical Revolution Peripheral Atherectomy System is a low-profile rotational atherectomy system (140,000 RPM) with continuous mechanical aspiration for below-the-knee (BTK) and above-the-knee (ATK) applications, including multiple plaque morphologies ranging from thrombus and soft plaque to severely calcified lesions and chronic total occlusions (CTOs). This device is operated by a 72 watt power supply similar in size to a laptop computer charger and requires no capital equipment and minimal setup time prior to use.
Jeffrey G. Carr, M.D., FACC, FSCAI, Tyler Cardiac and Endovascular Center, served as the study’s principal investigator and stated, “PAD and CLI are associated with significant patient quality of life and major health issues. The recent clearance of Revolution technology will assist the interventional community in the effective reduction of calcification and vessel preparation in multiple lesion morphologies prior to adjunctive therapy utilizing percutaneous transluminal angioplasty (PTA), drug-coated balloons and stenting. Continuous mechanical aspiration to reduce the potential for distal embolization is a distinct advantage associated with this technology. Intraprocedural embolization continues to be a key concern for many atherectomy procedures.”
Rex Medical President Lindsay L. Carter said the company anticipates obtaining CE Mark for the Revolution Peripheral Atherectomy System in the fourth quarter of 2019.
For more information: www.rexmedical.com


 November 08, 2024
November 08, 2024