The California Heart & Vascular Clinic in El Centro, Calif., treated the first post-FDA clearance patient with a DABRA system.
As debates about the current state and future of healthcare rage in Congress, the media and healthcare settings across the nation, there appears to be very little common ground upon which stakeholders agree. Yet common aspirations that are shared by most participants include better patient care, better clinical outcomes and lower costs.
One positive trend in the past six years in realizing these aspirations is the growth of peripheral vascular interventional (PVI) procedures that are performed in office-based labs (OBLs) instead of in hospitals. Specifically, the treatment of peripheral vascular disease (PAD), including atherectomy procedures, in the OBL represents an effective new treatment model that is beneficial not just for the patient but also for the entire healthcare system.
“Better for the patient” may be measured on efficacy, safety and patient satisfaction. Within patient satisfaction are embedded numerous other components, including continuity of care (for example, same provider, staff and surroundings), an office-based environment that is familiar and less intimidating than a hospital, and convenience and ease for family members and caregivers and the avoidance of nosocomial infection.
“Better for the healthcare system” is more than just a reduction in cost. This means fewer complications and follow-on procedures, both of which ultimately save money for the patient and healthcare system. It is also about better conditions for providers and creating fertile ground for innovation in devices, techniques and procedures – all toward the goal of bettering patient care and, again, lowering costs. In such optimized conditions, there may be faster adoption of new technologies, including devices and procedures that could deliver better health outcomes for patients.
As beneficial to patients and the healthcare system as the increase in OBL-based PVIs has been over recent years, critics continue to raise questions about the efficacy and safety of these procedures that are being performed outside of hospitals. The detractors raise concerns that many OBLs are in isolated areas, far from tertiary hospitals with surgeons on call. But the remote locations of some of these OBLs are, in fact, one of the key benefits of this trend, as these practices give access to patients who would otherwise not be able to get the treatment that they need, particularly those in rural areas and who face the risk of their diseases progressing toward amputation.
Nonetheless, critics claim that OBLs put patients at risk, as they are often too far away from hospitals, lack staff and resources to handle major complications and do not have adequate transfer options. They also assert that OBLs do not treat the sicker patients but instead “cherry pick” less compromised patients with less complex presentations, while patients with more serious co-morbidities and complex diseases continue to be treated in hospitals.
Through sharing data from my own OBL, along with a case that highlights many of the aforementioned advantages of PVI procedures that are being performed in OBLs, I can demonstrate the safety and efficacy on patients who are as compromised / sick as patients in hospitals. The case will also show the success with a patient who had been previously treated at a large, well-known tertiary hospital in which intervention failed and, in fact, created further complications.
This patient was referred to and treated in my OBL with the DABRA catheter and laser system, with great success. This case is a prime example of how OBLs are on the cutting edge of innovations in devices and techniques, as OBLs lead the adoption of new technologies, drive innovation by participating in clinical studies and are not slowed down by purchasing procedures and budget cycles of hospitals for capital equipment.
Beyond that, OBLs are not hampered by using devices that are proscribed by purchasing departments, which are removed from the actual procedures or by mandates to perform possibly unnecessary steps simply because they are in a residency training protocol. As each OBL’s operator is unencumbered by hospital-mandated bundling or group purchasing, the operator is free to choose whichever device he or she deems most appropriate – and in the patient’s best interest.
OBL Data Compares Favorably With Hospital Data for PVI
As a single operator in an OBL that is located 120 miles from a major city and without immediate access to a stand-by vascular surgeon, it is my conviction that complex peripheral vascular disease procedures can be performed in an outpatient setting with no compromise to patient safety, a low rate of complications and a high success rate.
While large-scale, prospective, multi-center studies, such as the DEFINITIVE LE study, demonstrated the safety and efficacy of atherectomy in hospitals, there has been scant outpatient data published and none with large-scale data from a single OBL. Therefore, I conducted a retrospective case note review of outpatient percutaneous PVD procedures that were performed at my lab between December 2012 and February 2016. Here is what I found.
Baseline characteristics of 767 patients who were included in my analysis:
• Claudicants: 57.4 percent had diabetes mellitus, 35.2 percent were smokers, 48.9 percent had hypertension, 31.5 percent had coronary heart disease, 37.0 percent had hyperlipidaemia and 16.1 percent had renal insufficiency.
• CLI patients: 66.8 percent had diabetes mellitus, 38.0 percent were smokers, 57.1 percent had hypertension, 35.5 percent had coronary heart disease, 36.8 percent had hyperlipidaemia and 21.1 percent had renal insufficiency.
• Total procedural success was 93.2 percent, with 95 percent success in complex lesions. The complication rate was 9.52 percent, with a total minor complication rate of 9 percent and only 0.52 percent total major complication rate.
This data demonstrates that complex percutaneous procedures for PVD can be performed safely in an outpatient setting with a low rate of complications. Minor complications are infrequent and do not require hospitalization. It further supports the fact that the cost savings and enhanced patient convenience/comfort of OBL-based PVD procedures are not at the expense of efficacy of the procedures or putting patients’ safety at risk.
Finally, to further compare the efficacy and safety of OBL PVD with those that are performed in hospitals, I benchmarked my data against the landmark DEFINITIVE LE study. As the chart below illustrates, with just slightly fewer (32) patients treated, my OBL treated sicker patients with a greater number of lesions, almost 200 percent the number of total occlusions with approximately one-third the major complications rate and a similar procedural success rate.
OBLs are not about choosing the easiest patients, OBLs are about performing the complex cases on the sickest and most compromised patients, with similar efficacy and safety to hospitals.
OBL Employs Cutting-Edge Technique to Save Tennis Player’s Leg
Here is an example of one case our lab performed. Jay S. is a 68-year-old male with a history of prosthetic endocarditis. He is a non-smoker who led a healthy lifestyle, including playing tennis almost daily until Fall 2016 when acute pain in his right leg prevented him from doing so. Jay had previously been a tennis champion in the Southern California 60+ age bracket.
On July 5, 2017, Jay had a thrombectomy performed with a Penumbra Cat6 at a well-known tertiary hospital in San Diego. After the procedure on his right leg, Jay was in an immense amount of pain anytime he tried to walk or get out of bed, and even at rest. The procedure had failed to recanalize the completely occluded right popliteal artery and instead created a dissection in the P2 section of the popliteal artery.
The interventionalist told him that he would never play tennis again and unless he developed adequate collateral vessels, he was on the road to amputation.
Jay was subsequently referred to my lab, and on Aug. 14, 2017, I did a diagnostic angiogram of the right leg by contralateral approach, which showed a large, eccentric linear dissection in the right infrapopliteal P2 segment of the popliteal artery. There was single vessel run-off to the foot via the right peroneal vessel, and the right AT was completely occluded with no demonstrable nub. The small nub of the right PT extended only about 2-3 mm, with no run off to the right foot.
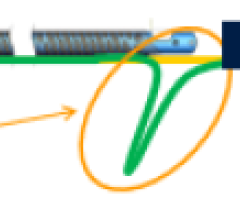
I initially attempted to cross the eccentric dissection at the level of the right popliteal artery to get into the true lumen using multiple wires, but I was unsuccessful. Every wire that I attempted went into the flap of the dissection.
At that point, I elected to use the laser system to ablate near the severely stenotic area in the P2 section of the popliteal artery. I made three passes with the system, with the help of a 6 French by 90 cm supportive sheath. The DABRA made the turn and stayed in the true lumen, making a small channel by ablating the plaque very close to dissection, without causing further dissection, in a textbook-perfect example of micro-plaque ablation.
The laser system performed accurate, precise plaque ablation around the dissection and resulted in a clear channel for laminar flow within the true lumen, enabling the 0.14 Glide wire to easily cross the critical stenosis at the level of the dissection.
With a support catheter, the totally occluded PT, which had a 2-3 mm nub, was crossed easily, and the wire was placed into distal PT, followed by a 3 x 200 mm-long POBA for three minutes. Next, a 3.5 x 40 mm long balloon was used to treat the ostium of the PT and the popliteal artery at 10 to 12 atmospheres for three minutes. Subsequently, the support catheter and wire were removed, and a small amount of contrast was injected that confirmed we were in the true lumen in the distal PT.
Post-procedure, the popliteal had less than 10 to 15 percent residual stenosis. The PT, which had been completely occluded, now had less than 10 percent residual stenosis and excellent runoff all of the way in-line flow to the foot via the PT with excellent flow in the plantar branches. The peroneal vessel, which previously had very poor flow, now had a brisk flow all of the way to the ankle. The AT was intermittently seen with a faint visualization of the right DP vessel.
After a minimum amount of time in recovery, Jay returned home. Two days later, on Aug. 16, 2017, Jay returned to playing tennis. His leg and tennis game have continued to improve, and he recently won the Men’s San Diego Under-70 Championship. This demonstrates the true power behind OBLs in treating PVI procedures, ultimately changing medicine as we know it today.
See learn more about the DABRA system in the VIDEO ”Editor's Choice of the Most Innovative New Technologies at TCT 2017.”
Editor’s note: Athar Ansari, M.D., FACC, director of the California Heart and Vascular Clinic in El Centro, Calif., is board certified in general cardiology, interventional cardiology and endovascular disease. He is a pioneer in minimally invasive outpatient-based treatment for peripheral artery disease. He was an investigator in DABRA’s pivotal study, treated the first post-FDA clearance DABRA patient, and performed the first ever DABRA System case on arterial blockages via brachial artery access.
Editor’s note: The DABRA system is only indicated for atherectomy in Europe. Ra Medical Systems said it is working with the FDA to get this indication.


 November 08, 2024
November 08, 2024