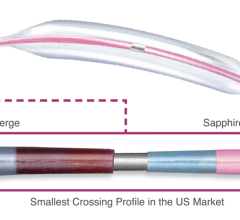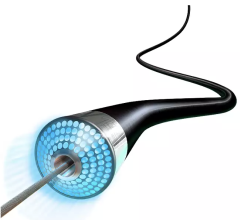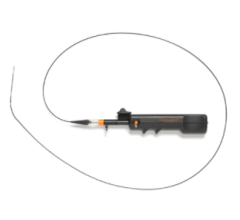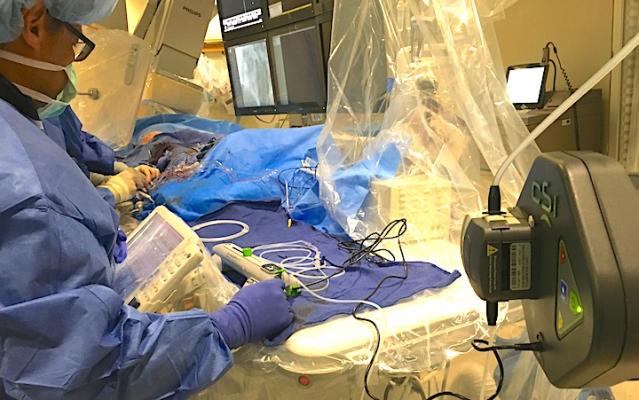
A CSI Diamondabck 360 atheretectomy system in use during a cath lab procedure at Henry Ford Hospital in Detroit. Photo by Dave Fornell
February 13, 2020 — The U.S. Food and Drug Administration (FDA) has issued its final guidance on "Peripheral Vascular Atherectomy Devices – Premarket Notification [510(k)] Submissions."
The guidance applies to atherectomy devices used in the peripheral vasculature, which are classified by the FDA as class II devices. Atherectomy devices used in coronary vasculature are outside the scope of this final guidance.
This guidance document provides recommendations for 510(k) submissions for peripheral artery disease (PAD) atherectomy devices. The FDA said the recommendations reflect current review practices and are intended to promote consistency and facilitate efficient review of peripheral vascular
atherectomy submissions.
This guidance is intended to help manufacturers evaluate their device, enhance consistency of performance testing, and promote the safe use of these devices in clinical settings.
Based on comments received on the draft guidance, the FDA said the final guidance includes additional clarifying details, including examples, about content related to corrosion testing, bench testing, and software.


 November 08, 2024
November 08, 2024