August 19, 2019 — The U.S. Food and Drug Administration (FDA) granted market clearance the Barostim Neo System for the ...
Pacemakers
This channel includes news and new technology innovations for pacemakers used to treat bradycardia.
July 31, 2019 — The chances of patients experiencing complications after having a cardiac device implanted vary ...
June 4, 2019 — Orchestra BioMed Inc. announced the presentation of two-year clinical data from the European Moderato I ...

May 15, 2019 — The Heart Rhythm Society (HRS) had 21 late-breaking study presentations at the 2019 Heart Rhythm ...
May 15, 2019 — A new infection risk scoring system has been developed based on data from the large PADIT Trial.[1] The ...
May 14, 2019 – Results from new research show that passengers with cardiac implantable electronic devices (CIEDs), such ...
May 13, 2019 – Results from a new survey are the first to report a large discrepancy in patient’s knowledge of their ...
May 7, 2019 — The U.S. Food and Drug Administration (FDA) issued a safety communication to alert healthcare providers ...
March 29, 2019 — A research team from Imperial College London believes a new software could speed up the diagnosis and ...
Khaldoun Tarakji, M.D., MPH, associate section head, section of electrophysiology and pacing in the Robert and Suzanne ...
March 21, 2019 — The U.S. Food and Drug Administration (FDA) approved Impulse Dynamics’ Optimizer Smart system for ...
February 20, 2019 — Medtronic is recalling its dual chamber implantable pulse generators (IPGs) due to the possibility ...

To extract or abandon broken or infected implantable, venous electrophysiology (EP) device leads has been a debate for ...
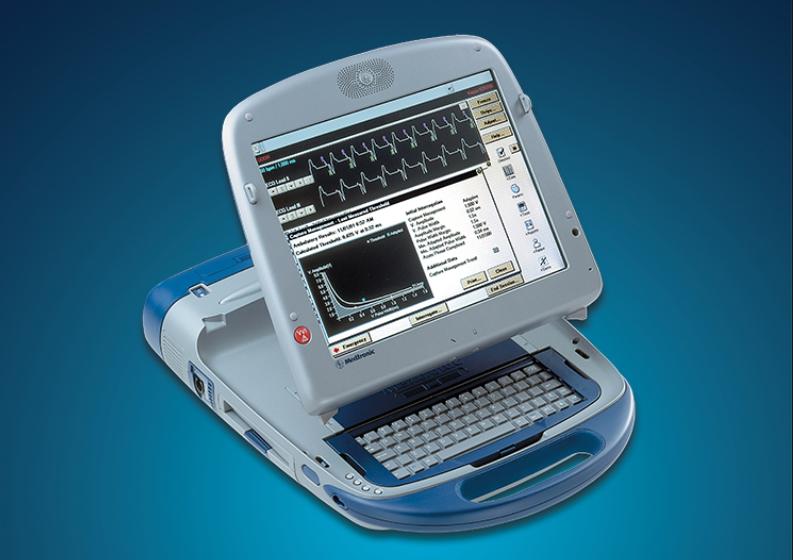
October 17, 2018 — The U.S. Food and Drug Administration (FDA) has reviewed information about potential cybersecurity ...
October 2, 2018 — Two-year results of the Moderato I Study demonstrated immediate, substantial and sustained reduction ...

 August 19, 2019
August 19, 2019
![A new infection risk scoring system has been developed based on data from the large PADIT Trial.[1] The new scoring system was presented as a follow up to that study during a late-breaking session at Heart Rhythm 2019, the Heart Rhythm Society's 40th Annual Scientific Sessions.](/sites/default/files/styles/content_feed_medium/public/PADIT_Infection_Risk_score.jpg?itok=O1-YAcMm)