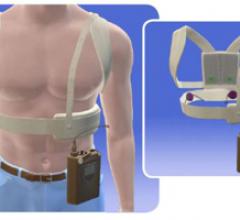
The Arrow AutoCAT 2 WAVE Interaotic balloon pump (IABP) System provides counterpulsation support…
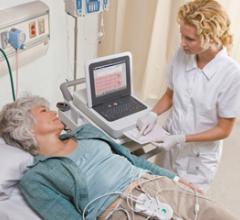
September 9, 2009 – Royal Philips Electronics unveiled its PageWriter TC50 cardiograph, which…
September 8, 2009 – Actelion Pharmaceuticals U.S. Inc. recently announced the first commercial…
The HI-TORQUE Balance Middle Weight (BMW) Universal II Guide Wire offers the same performance of…
The Perclose ProGlide 6 Fr. Suture-Mediated Closure System delivers a secure, reliable vessel…
September 3, 2009 – ZOLL Medical Corp. received approval from the FDA to market and sell a new…
The HI-TORQUE Versacore .035 Guide Wire is a peripheral guide wire is designed for routine…
The FoxCross PTA Catheter is a next-generation .035 balloon dilatation catheter is used to open…
September 2, 2009 – A new analysis from the JUPITER study showed CRESTOR (rosuvastatin calcium…
August 21, 2009 – Royal Philips Electronics today announced the release of the Alice PDx…
August 21, 2009 – 3M today introduced the 3M Littmann Electronic Stethoscope Model 3200, a next-…
August 18, 2009 – Cardiva Medical Inc. recently received FDA clearance for its Cardiva Catalyst…
August 17, 2009 - Reverse Medical Corporation announced that it has received U.S. FDA 510k…
August 14, 2009 – Philips is currently introducing its Xcelera R3.1 cardiology informatics…
August, 13, 2009 – Atherotech Inc. this week added two new cardiometabolic tests – the PLAC Test…
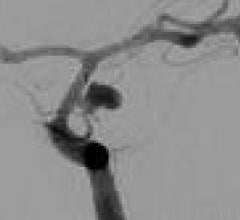
August 11, 2009 – Pathway Medical Technologies Inc. today introduced the 7 French compatible…
August 3, 2009 – Kowa Research Institute (KRI) and Kowa Pharmaceuticals America Inc. (KPA) said…
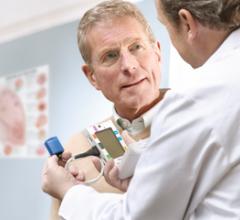
July 31, 2009 – Heart Force Medical Inc. this week received FDA clearance for its digital…
July 31, 2009 – United Therapeutics Corp. said yesterday the FDA approved TYVASO (treprostinil)…
July 27, 2009 – Carestream Health Inc. today launched a new Web-based cardiology PACS that…


 September 09, 2009
September 09, 2009