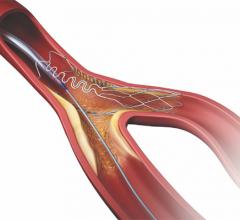
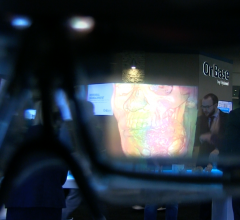March 20, 2017 — Teleflex Inc. recently announced 510(k) clearance by the U.S.
March 20, 2017 — Agfa HealthCare announced the release a new version of its…
March 20, 2017 — Cardiologists can now access the advanced…
Medtronic plc announced U.S. Food and Drug Administration (FDA) 510(k) clearance for its Reveal…
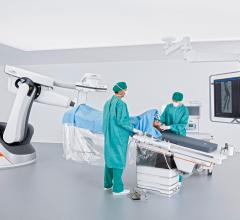
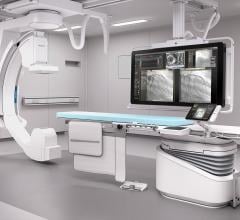
The U.S. Food and Drug Administration (FDA) has cleared the Artis pheno robotic C-arm…
March 14, 2017 — The United States Food and Drug Administration (FDA) has expanded use of the…
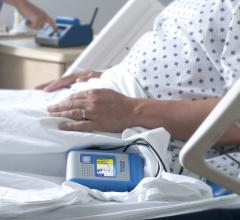
Zoll Medical Corp. announced that the company’s Hospital Wearable Defibrillator (HWD) has been…
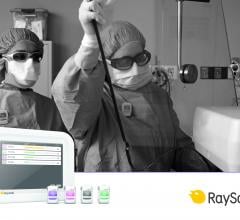
March 7, 2017 — Unfors RaySafe recently introduced the RaySafe i3, to its suite of real-time…
ON Semiconductor announced the release of a new CCD image sensor that enables video imaging…
March 6, 2017 – The U.S.
Philips recently announced the global launch of Azurion, its next-generation image-guided…
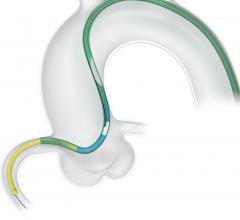
Teleflex Inc. has announced 510(k) clearance by the U.S. Food and Drug Administration (FDA) and…
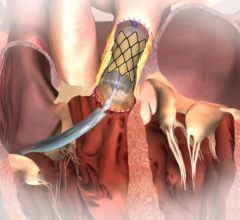
March 2, 2017 — The U.S. Food and Drug Administration (FDA) cleared CeloNova BioSciences Inc.
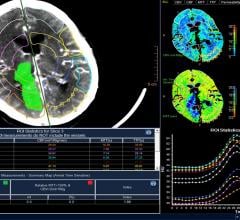
EchoPixel recently announced True 3-D print support functionality, a new set of software tools…
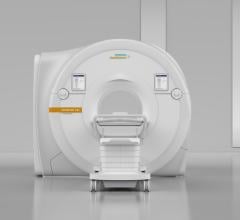
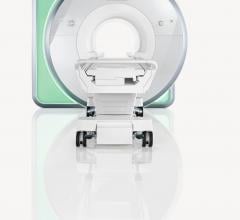
February 28, 2017 — Magnetom Vida, the new high-end 3 Tesla magnetic resonance imaging (MRI)…
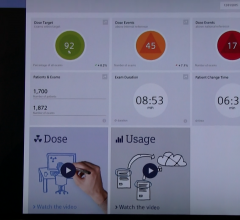
At the 55th Annual Meeting for the Health Information and Management Systems Society (HIMSS),…
TeraRecon debuted their new high-performance, cloud-based augmented reality solution, the…
Philips recently announced the introduction of IntelliSpace Enterprise Edition at the 2017…
Siemens Healthineers announced that the U.S. Food and Drug Administration (FDA) has cleared the…

 March 20, 2017
March 20, 2017