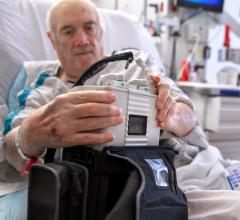
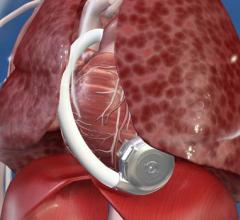
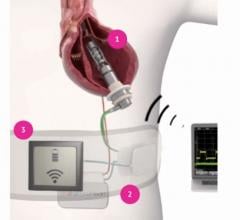
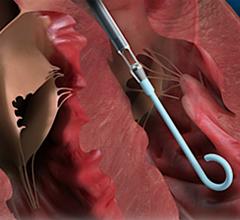
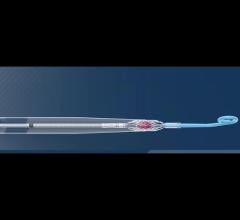
While many cardiac and vascular procedures have largely moved to minimally invasive techniques, the size of these ...
Ventricular Assist Devices (VAD)
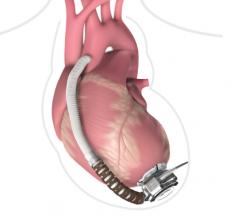
This channel includes news and new technology innovations for ventricular assist devices (VAD). VADs are a type of mechanical hemodynamic support device that helps increase blood flow in people who have ventricles that are not work properly due to heart failure, cardiogenic shock, cardiomyopathy or myocardial infarction. Most often these devices support the left ventricle, so they are often referred to as left ventricular assist devices (LVAD). VADS come in two types, surgically implanted, usually as a bridge to heart transplant, and percutanenous catheter-based pumps used for temporary hemodynamic support. Examples of temporary percutaneous pumps include the Impella and TandemHeart devices.
March 2, 2021 — Penn State Health Milton S. Hershey Medical Center became the second hospital in the nation to implant a ...
March 1, 2021 — Medtronic is recalling the HeartWare Ventricular Assist Device (HVAD) Pump Implant Kit because the left ...
January 13, 2021 — FineHeart, a preclinical-stage medical device company developing a novel left ventricular assist ...
December 21, 2020 — U.S. Food and Drug Administration (FDA) approved updated labeling December 17 for Abbott's HeartMate ...
December 2, 2020 — The Centers for Medicare and Medicaid Services (CMS) finalized updates to Medicare coverage policies ...
Chuck Simonton, M.D., chief medical officer at Abiomed, discusses some of the new technologies and clinical trials the ...
October 16, 2020 — The Restore EF Study demonstrates the use of contemporary best practices, including attempting a more ...

August 21, 2020 — The U.S. Centers for Medicare and Medicaid Services (CMS) proposed updates to coverage policies for to ...
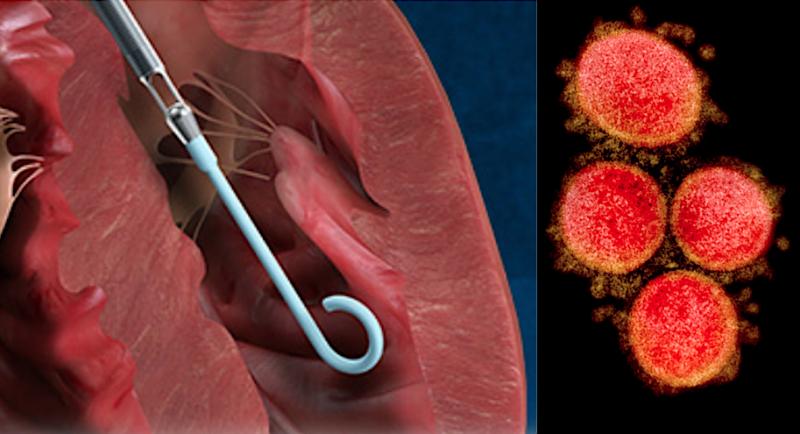
August 5, 2020 — The U.S. Food and Drug Administration (FDA) issued an emergency use authorization (EUA) this week for ...
June 5, 2020 — Abiomed announced the U.S. Food and Drug Administration (FDA) has approved the company's investigational ...
This is a quick animation demonstrating how the new 9 French Abiomed Impella ECP expands to approximately 18 French and ...
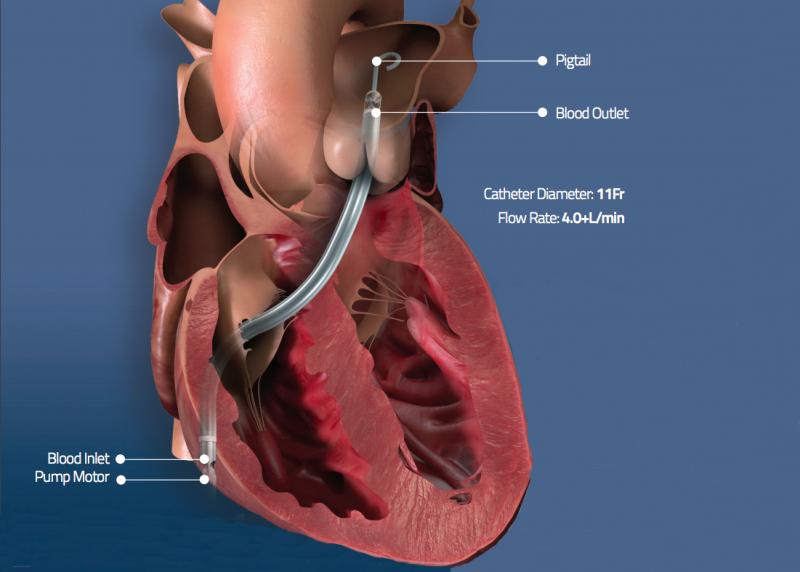
June 1, 2020 — The U.S. Food and Drug Administration (FDA) has issued an emergency use authorization (EUA) for the Abiom ...
May 29, 2020 — Medtronic is recalling its HeartWare HVAD left ventricular assist device (LVAD) pump outflow graft and ...
May 27, 2020 — Carmat, a developer of the of a next generation advanced total artificial heart, announces the first ...


 March 09, 2021
March 09, 2021